��Ŀ����
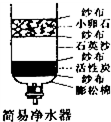 ijУ�о���ѧϰС��Ը���ij���ں�ˮ�ʵ�״��������صĵ����о���
ijУ�о���ѧϰС��Ը���ij���ں�ˮ�ʵ�״��������صĵ����о���
��1����ͼ��ʾ�����ƾ�ˮ���е�______���ʣ��ɳ�ȥˮ�е���ζ��ɫ�أ�
��2��Ӳˮ���������������������鷳����������Ӳˮ���ܻ��������ڽ�ʯ������Ӳˮϴ��������˷ѷ�����ϴ���ɾ���ʱ�䳤�˻���ʹ�����Ӳ��ͨ����ѧѧϰ������֪������______�����ָ��ںӵ�ˮ����Ӳˮ������ˮ��������Ҫ����ˮ��Ӳ�ȣ��ɲ��õķ�����______��
��3������Ϊ������Ϊ������ں�ˮ���½�����______�����ţ���
���� A���ںӱ���ֲ������ľ�� B������������ˮֱ�������ںӡ�C�������������ں���
��4������ˮ����ָ������ˮ�����ﵽ�涨��ˮ�ʱ�������һ����Χ���ٴ�ʹ�õķ�����ˮ���������˽�ˮ�ľ���������д����������ˮ���������ɡ���ˮ���Ĺ����У������õ����ַ���______��______�����á���ˮ����ʵ��������______��
��5��X��һ�����͵�����ˮ����������ҵ����ȡX �Ļ�ѧ����ʽΪ��Cl2+2NaClO2=2NaCl+2X����X�Ļ�ѧʽΪ______��
�⣺
��1�����û���̿�������ԣ��ɳ�ȥˮ�е���ζ��ɫ�أ�
��2��Ӳˮ�к��н϶�ơ�þ���ӣ���Ӱ����������ݣ��ڶ�ˮ�������ʱ��ˮ�иơ�þ���ӻ��γɳ���������ˮ�����Ӻ�������ˣ�ͨ��ʹ�÷���ˮ����ˮ����Ӳ����ͨ����еķ�������ˮ��Ӳ�ȣ�
��3����������ˮ�����������ˮ�У���������ں�ˮ���½���
��4����������ˮ���������ɡ���ˮ���Ĺ����У���Ҫ��ȥˮ�е�һЩ���岻���P��ζ�������Ҫ���й��˺�������������ˮ����һ����Χ���ٴ�ʹ�ã���˿ɽ�Լˮ��Դ��������ˮ�ŷţ�������Ⱦ����
��5���۲췽��ʽ��֪��Cl2+2NaClO2=2NaCl+2X����Ӧǰ��4����ԭ�ӣ�2����ԭ�ӣ�4����ԭ�ӣ���Ӧ����2����ԭ�ӣ�2����ԭ�ӣ�����1��X������1����ԭ�Ӻ�2����ԭ�ӹ��ɣ���ѧʽΪ��ClO2��
�ʴ�Ϊ��
��1������̿��
��2������ˮ����У�
��3��BC��
��4�����ˣ���������Լˮ��Դ���������ˮ�ŷţ�������Ⱦ����
��5��ClO2��
��������1�����ݾ���ˮ��Ҫ��ȥˮ�е���ζ��ɫ�أ�ͨ���û���̿��
��2������Ӳˮ����ˮ�IJ�ͬ���ж�����Ӳˮ����ˮ������Լ�������ˮ��Ӳ�ȿɲ��õķ�����
��3������ˮ��Ⱦ�����֪ʶ���жϻ�����ں�ˮ���½�����Ϊ��
��4��������ˮ���ص㣬�жϽ�������ˮ���������ɡ���ˮ���Ĺ����в��õľ��������������������������壻
��5�����ݻ�ѧ��Ӧǰ��ԭ�ӵ��������Ŀ����������
�������˽�ˮ����Ⱦ������ˮ�ľ������õIJ�����������ˮԭ���������غ㶨�ɵ�Ӧ�ã����ǽ����������߱��Ļ�����
��1�����û���̿�������ԣ��ɳ�ȥˮ�е���ζ��ɫ�أ�
��2��Ӳˮ�к��н϶�ơ�þ���ӣ���Ӱ����������ݣ��ڶ�ˮ�������ʱ��ˮ�иơ�þ���ӻ��γɳ���������ˮ�����Ӻ�������ˣ�ͨ��ʹ�÷���ˮ����ˮ����Ӳ����ͨ����еķ�������ˮ��Ӳ�ȣ�
��3����������ˮ�����������ˮ�У���������ں�ˮ���½���
��4����������ˮ���������ɡ���ˮ���Ĺ����У���Ҫ��ȥˮ�е�һЩ���岻���P��ζ�������Ҫ���й��˺�������������ˮ����һ����Χ���ٴ�ʹ�ã���˿ɽ�Լˮ��Դ��������ˮ�ŷţ�������Ⱦ����
��5���۲췽��ʽ��֪��Cl2+2NaClO2=2NaCl+2X����Ӧǰ��4����ԭ�ӣ�2����ԭ�ӣ�4����ԭ�ӣ���Ӧ����2����ԭ�ӣ�2����ԭ�ӣ�����1��X������1����ԭ�Ӻ�2����ԭ�ӹ��ɣ���ѧʽΪ��ClO2��
�ʴ�Ϊ��
��1������̿��
��2������ˮ����У�
��3��BC��
��4�����ˣ���������Լˮ��Դ���������ˮ�ŷţ�������Ⱦ����
��5��ClO2��
��������1�����ݾ���ˮ��Ҫ��ȥˮ�е���ζ��ɫ�أ�ͨ���û���̿��
��2������Ӳˮ����ˮ�IJ�ͬ���ж�����Ӳˮ����ˮ������Լ�������ˮ��Ӳ�ȿɲ��õķ�����
��3������ˮ��Ⱦ�����֪ʶ���жϻ�����ں�ˮ���½�����Ϊ��
��4��������ˮ���ص㣬�жϽ�������ˮ���������ɡ���ˮ���Ĺ����в��õľ��������������������������壻
��5�����ݻ�ѧ��Ӧǰ��ԭ�ӵ��������Ŀ����������
�������˽�ˮ����Ⱦ������ˮ�ľ������õIJ�����������ˮԭ���������غ㶨�ɵ�Ӧ�ã����ǽ����������߱��Ļ�����

��ϰ��ϵ�д�
 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�
�����Ŀ
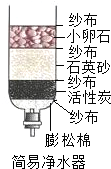 ijУ�о���ѧϰС��Ը���ij���ں�ˮ�ʵ�״��������صĵ����о���
ijУ�о���ѧϰС��Ը���ij���ں�ˮ�ʵ�״��������صĵ����о���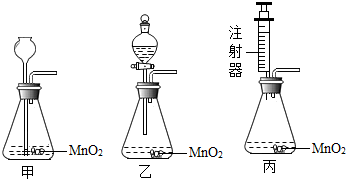 ��2010?���ݣ�ijУ�о���ѧϰС��Բ��÷ֽ����������Һ��ȡ����������ʵ��̽����
��2010?���ݣ�ijУ�о���ѧϰС��Բ��÷ֽ����������Һ��ȡ����������ʵ��̽����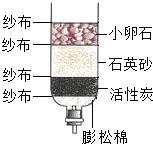 ijУ�о���ѧϰС��Ժ���ij��ˮ�ʵ�״��������صĵ����о���
ijУ�о���ѧϰС��Ժ���ij��ˮ�ʵ�״��������صĵ����о��� ijУ�о���ѧϰС��Ը���ij���ں�ˮ�ʵ�״��������صĵ����о���
ijУ�о���ѧϰС��Ը���ij���ں�ˮ�ʵ�״��������صĵ����о���