��Ŀ����
����Ŀ����10�֣�̼Ԫ��������������ʵĻ���Ԫ�ء�
��1�����飨CH4����һ�������Դ���ڼ�����̼��Ԫ�������� ��0.1mol���� ��������ӣ�����Ϊ g��д��������ȫȼ�յĻ�ѧ����ʽ ��
��2�������±�����ú��ȣ�����Ȼ����ȼ�ϵ��ŵ��� ��
1g������ȫȼ�� | ||
����������̼������/g | �ų�������/kJ | |
���� | 2.75 | 56 |
̼ | 3.67 | 32 |
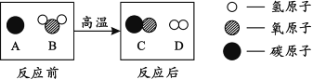
��3��Ϊ���ú�������ʣ��ɽ���ת��Ϊ��ȼ�����壬�÷�Ӧ����ʾ��ͼ���£���������ķ��Ӹ�����Ϊ ��
��4����ʯȼ����Ҫ����ú�� ����Ȼ����
��5���ܶ���Ȼ��ʯ�к���̼Ԫ�أ����̿����Ҫ�ɷ���̼����(MnCO3)��������Ԫ�صĻ��ϼ�Ϊ ��
��6��Һ̬������̼�������˾ȵ����ҷ����Ļ��֣�����˵����ȷ���� ������ĸ��ţ���
A��Һ̬������̼��������Ⱦ����
B��������̼�ɸ����ڿ�ȼ����棬��������
C��Һ̬������̼����ʱ���ȣ����Ϳ�ȼ����Ż��
���𰸡���1��3��1 6.02��1022 1.6 CH4 + 2O2��ȼCO2 + 2H2O
��2������������Ȼ����ú��ȫȼ�գ���Ȼ��ȼ�ղ���������̼����������ú���ų�����������ú
��3��1:1 ��4��ʯ�� ��5��+ 2 ��6��AB
��������
�����������1�����飨CH4����̼��Ԫ��������=12:1��4=3��1��1mol���������к��еķ�����6.02��1023����0.1mol����6.02��1022������=0.1mol��16g/ mol=1.6 g��������ȫȼ�յĻ�ѧ����ʽΪ��CH4 + 2O2��ȼCO2 + 2H2O
��2�������������ݷ�������ú��ȣ�����Ȼ����ȼ�ϵ��ŵ�������������Ȼ����ú��ȫȼ�գ���Ȼ��ȼ�ղ���������̼����������ú���ų�����������ú
��3�����ݷ�Ӧ����ʾ��ͼ����д����ѧ����ʽ��H2O + C ���� H2 + CO����������ķ��Ӹ�����1:1 ��4����ʯȼ����Ҫ����ú��ʯ������Ȼ��
��5������Ԫ�ػ��ϼ۵�һ����ɣ��ڻ������У��������ϼ۵Ĵ�����Ϊ0����̼����(MnCO3)�У�CO3�Ļ��ϼ�Ϊ-2������Ԫ�صĻ��ϼ�+ 2
��6��A��Һ̬������̼��������Ⱦ��������ȷ��B��������̼�ɸ����ڿ�ȼ����棬������������ȷ��C��Һ̬������̼����ʱ���ȣ������¶ȣ�ʹ�¶ȵ��ڿ�ȼ����Ż�㣬�����ǽ����Ż�㣬��ѡ��AB

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�