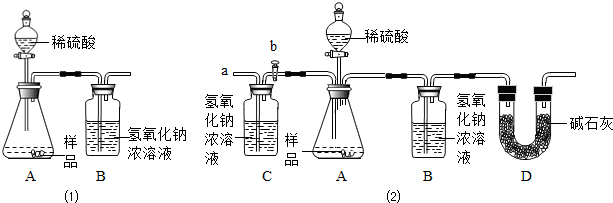
��Ŀ����
��2013?̩���������ӽǿ����ʵı仯�ǻ�ѧ���е�˼ά��ʽ����������ʾ��ͼ�ֱ��ʾ�����Ȼ��ƵIJ�ͬ��ѧ��Ӧ�������ͼʾ�ش�������⣺
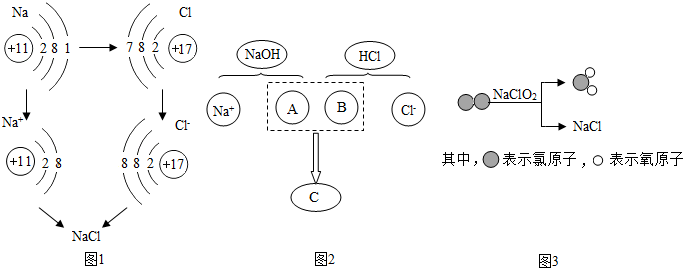
��1��ͼ1�ǽ�������������Ӧ�����Ȼ��Ƶ���ʾ��ͼ����ͼ1��֪��Ԫ�صĻ�ѧ������
A������������ B���ڲ����������C�����Ӳ���
��2��ͼ2������NaOH��Һ�����ᷴӦ����ʵ�ʣ�ͼ��A��B��C��Ӧ����Ļ�ѧʽ�����ӷ�������Ϊ
��3��ͼ3��ʾ��Ӧ�Ļ�ѧ����ʽΪ

��1��ͼ1�ǽ�������������Ӧ�����Ȼ��Ƶ���ʾ��ͼ����ͼ1��֪��Ԫ�صĻ�ѧ������
A
A
������ĸ��ţ������й�ϵ��A������������ B���ڲ����������C�����Ӳ���
��2��ͼ2������NaOH��Һ�����ᷴӦ����ʵ�ʣ�ͼ��A��B��C��Ӧ����Ļ�ѧʽ�����ӷ�������Ϊ
OH-��H+��H2O
OH-��H+��H2O
����3��ͼ3��ʾ��Ӧ�Ļ�ѧ����ʽΪ
Cl2+2NaClO2=2ClO2+2NaCl
Cl2+2NaClO2=2ClO2+2NaCl
����������1�����ݷ�Ӧ��ԭ�ӵĺ�����ӱ仯����������
��2�������кͷ�Ӧ��ʵ�ʶԻ�ѧʽ��������������жϼ��ɣ�
��3��������ʾ��ͼ����ط��ӵĽṹ��д��صĻ�ѧʽ��������д�йصķ���ʽ���ɣ�
��2�������кͷ�Ӧ��ʵ�ʶԻ�ѧʽ��������������жϼ��ɣ�
��3��������ʾ��ͼ����ط��ӵĽṹ��д��صĻ�ѧʽ��������д�йصķ���ʽ���ɣ�
����⣺��1�����ݽ�������������Ӧ�����Ȼ��Ƶ���ʾ��ͼ��֪���ñ仯����������ԭ�ӵ������������������˱仯�����Կ�֪Ԫ�صĻ�ѧ������ԭ�ӵ����������������й�ϵ��
��2���������������ܹ�����������Ӻ����������ӣ������ܹ�����������Ӻ������ӣ�����A��Ӧ��������������B���������ӣ��������ӽ���ܹ�����ˮ���ӣ�����C��Ӧ����ˮ���ӣ�
��3����ͼʾ�з��ӵ��۽ṹ��֪�÷�Ӧ�ķ�Ӧ����Cl2��NaClO2����������ClO2��NaCl�����Ը÷�Ӧ�ķ���ʽΪCl2+2NaClO2=2ClO2+2NaCl��
�ʴ�Ϊ����1��A����2��OH-��H+��H2O����3��Cl2+2NaClO2=2ClO2+2NaCl��
��2���������������ܹ�����������Ӻ����������ӣ������ܹ�����������Ӻ������ӣ�����A��Ӧ��������������B���������ӣ��������ӽ���ܹ�����ˮ���ӣ�����C��Ӧ����ˮ���ӣ�
��3����ͼʾ�з��ӵ��۽ṹ��֪�÷�Ӧ�ķ�Ӧ����Cl2��NaClO2����������ClO2��NaCl�����Ը÷�Ӧ�ķ���ʽΪCl2+2NaClO2=2ClO2+2NaCl��
�ʴ�Ϊ����1��A����2��OH-��H+��H2O����3��Cl2+2NaClO2=2ClO2+2NaCl��
�����������Ƕ����ʼ䷴Ӧ����֪ʶ�Ŀ��飬����Ĺؼ�����ȷ��ͼʾ���йغ��������йصķ��ӽṹ�����

��ϰ��ϵ�д�
�����Ŀ
 ��2013?̩������ͼ��A��B��C���ֹ������ʵ��ܽ�����ߣ����з�������ȷ���ǣ�������
��2013?̩������ͼ��A��B��C���ֹ������ʵ��ܽ�����ߣ����з�������ȷ���ǣ�������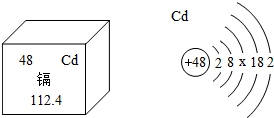 ��2013?̩�������꣬���ݡ��Ӵ��ס��¼��ٴ����������Ƕ�ʳƷ��ȫ��������Ⱦ�����˼����С��ͬѧ�������ӵ������Ϣ������Ԫ��λ�ڵ������ڢ�B�壮����������ɫ�й���Ľ������۵�320.9�棬�е�765�棬�ܶ�8.64g/cm3�������Ժ���չ�ԣ����ڳ�ʪ�����л���������ʧȥ���������ӿ������ᣬ�������ڼ���Ӷ�����ʮ���к��������������Ⱦ��ʳƷ��ˮ��������κ����༲�����ش��������⣺
��2013?̩�������꣬���ݡ��Ӵ��ס��¼��ٴ����������Ƕ�ʳƷ��ȫ��������Ⱦ�����˼����С��ͬѧ�������ӵ������Ϣ������Ԫ��λ�ڵ������ڢ�B�壮����������ɫ�й���Ľ������۵�320.9�棬�е�765�棬�ܶ�8.64g/cm3�������Ժ���չ�ԣ����ڳ�ʪ�����л���������ʧȥ���������ӿ������ᣬ�������ڼ���Ӷ�����ʮ���к��������������Ⱦ��ʳƷ��ˮ��������κ����༲�����ش��������⣺