��Ŀ����
��6�֣�ͭ���ڳ�ʪ�Ŀ������������⡣ͭ�⣨�׳�ͭ�̣�����Ҫ�ɷ���Cu2(OH)2CO3����ô��ͭ��ʲô���������������أ�Ϊ�ˣ�С��ͬѧ���������ʵ�飬�������̽����
��������衿ͭ������ͭ�������� �� ��ͬ���õĽ����
����Ʒ�����Ϊ��֤���裬С��ͬѧ���������ʵ�鷽����
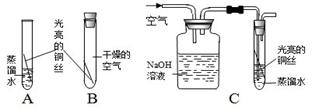
��1��B��ʵ��Ŀ���� ��
��2��װ��C����һ�����Դ����������������Ը��� ��NaOH��Һ�������ǣ��û�ѧ����ʽ��ʾ�� ��
��3��С��ͬѧ�����ʵ�鷽��������������Ҫ�ٲ���һ��ʵ�顣������ͼ���л����ʵ���װ��ͼ��

�����ۡ�����̽������֪��ͭ�����������
��������衿ͭ������ͭ�������� �� ��ͬ���õĽ����
����Ʒ�����Ϊ��֤���裬С��ͬѧ���������ʵ�鷽����

��1��B��ʵ��Ŀ���� ��
��2��װ��C����һ�����Դ����������������Ը��� ��NaOH��Һ�������ǣ��û�ѧ����ʽ��ʾ�� ��
��3��С��ͬѧ�����ʵ�鷽��������������Ҫ�ٲ���һ��ʵ�顣������ͼ���л����ʵ���װ��ͼ��

�����ۡ�����̽������֪��ͭ�����������
��������衿 H2O CO2
��1����֤ͭ��������������̼�Ӵ��Ƿ�����
��2��ͨ������ĵ�����Ӧ����NaOH��Һ�У�2NaOH + CO2="=" Na2CO3 + H2O
��3������ͼ
��1����֤ͭ��������������̼�Ӵ��Ƿ�����
��2��ͨ������ĵ�����Ӧ����NaOH��Һ�У�2NaOH + CO2="=" Na2CO3 + H2O
��3������ͼ

�������������ͭ�̵Ļ�ѧʽ�����������غ㶨��֪��������衿ͭ������ͭ��������������̼�� ˮ ��ͬ���õĽ��������Ʒ�������1��B��ʵ��Ŀ���Ǹ��ݶԱȵ�������֪��1����֤ͭ��������������̼�Ӵ��Ƿ����⣻��2��װ��C����һ�����Դ�����ͨ������ĵ�����Ӧ����NaOH��Һ�У�NaOH��Һ�������ǣ��û�ѧ����ʽ��ʾ��2NaOH + CO2="=" Na2CO3 + H2O����3��С��ͬѧ�����ʵ�鷽��������������Ҫ�ٲ���һ��ʵ��


��ϰ��ϵ�д�
 �ŵ������ϵ�д�
�ŵ������ϵ�д�
�����Ŀ

 Na2CO3+H2O+CO2��
Na2CO3+H2O+CO2��