��Ŀ����
ijУ��ѧ�о���ѧϰС���˽��ijʯ�ij��¹���һ��ʯ��ʯԭ�ϣ������������ʼȲ�����ˮ��Ҳ���������ᣬ����ʱҲ��������Ӧ����С��ȡ��4g��Ʒ���ⶨ��ʯ��ʯ�Ĵ��ȣ���δ֪��������������ϡ����40g��4�μ��룮ÿ�γ�ַ�Ӧ�����ˡ�����Ȳ���������������ʵ�������ÿ��ϡ�����������ʣ�������������±���������ˡ�����Ȳ������̣�������������������ģ���| ϡ��������� | ʣ���������� |
| ��һ�μ���10g | 3.0g |
| �ڶ��μ���10g | 2.0g |
| �������10g | 1.0g |
| ���Ĵμ���10g | 0.4g |
��2�����õ�ϡ���������ʵ����������Ƕ��٣�
��3�������������ʯ��ʯԭ�������������ʵ���ʯ������Ϊ100t������Ҫ����ʯ��ʯԭ������Ϊ���ٶ֣�����������ȷ��0.1��
��������1��ʯ��ʯ��Ʒǰ����ٵ�����Ϊ̼��Ƶ����������ʣ���0.4gΪ���ʵ��������ɴ˿����ʯ��ʯ��Ʒ��̼��Ƶ�����������
��2����������ʵ���֪ÿ10��ϡ������1��̼���ǡ����ȫ��Ӧ������̼��Ƶ��������10��ϡ���������ʵ��������������ϡ���������ʵ�����������
��3��������ʯ�ҵ����������ʯ��ʯ��������
��2����������ʵ���֪ÿ10��ϡ������1��̼���ǡ����ȫ��Ӧ������̼��Ƶ��������10��ϡ���������ʵ��������������ϡ���������ʵ�����������
��3��������ʯ�ҵ����������ʯ��ʯ��������
����⣺��1��ʯ��ʯ��Ʒ��CaCO3����������Ϊ��
��100%=90%
��2���⣺���һ��ʵ���У��������10g���������ʵ�����ΪX
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 73
1g X
=
x=0.73g
ϡ���������ʵ���������=
��100%=7.3%
��3���跴Ӧ��ʯ��ʯ������Ϊy������ʯ��ʯ�Ĵ��ȣ��������ʵ�����Ϊ10%y��
CaCO3
CaO+CO2��
100 56
90%y 100t-10%y
=
y=165.6t
��ʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ90%��ϡ���������ʵ���������Ϊ7.3%����������ʯ��ʯԭ�������������ʵ���ʯ������Ϊ100t������Ҫ����ʯ��ʯԭ������Ϊ165.6t��
| 4g-0.4g |
| 4g |
��2���⣺���һ��ʵ���У��������10g���������ʵ�����ΪX
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 73
1g X
| 100 |
| 1g |
| 73 |
| X |
x=0.73g
ϡ���������ʵ���������=
| 0.73g |
| 10g |
��3���跴Ӧ��ʯ��ʯ������Ϊy������ʯ��ʯ�Ĵ��ȣ��������ʵ�����Ϊ10%y��
CaCO3
| ||
100 56
90%y 100t-10%y
| 100 |
| 90%y |
| 56 |
| 100t-10%y |
y=165.6t
��ʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ90%��ϡ���������ʵ���������Ϊ7.3%����������ʯ��ʯԭ�������������ʵ���ʯ������Ϊ100t������Ҫ����ʯ��ʯԭ������Ϊ165.6t��
����������������ʱ�������õ���Ӧǰ�����������һ��Ҫ����Ӧǰ�����������ʲô�����������壬װ�õ�������ǰ����ٵ�һ��Ϊ�������������������ǰ����ٵ�һ��Ϊ�μӷ�Ӧ�Ĺ������ʵ�������

��ϰ��ϵ�д�
�����Ŀ
Ϊ�˲ⶨijƷ��ʳ�ô�����̼���Ƶ�����������ijУ��ѧ�о���ѧϰС���̽���������£�
[�������]��Ʒ��̼���Ƶ����������Ƕ��٣�
[֪ʶ��]
ʳ�ô������Ҫ�ɷ���̼���ƣ���������������Ȼ��ƣ���Ӧ�����в�����ˮ���Ȼ���Ļӷ���
[��Ʒ�����ʵ��]
����ͬѧ����ȡ12.00��Ʒ����ˮ�����Һ������Һ�м����������ʯ��ˮ�����ˡ�ϴ�ӡ�������õ���ɫ����10.00g��
����ͬѧ����ȡ12.00��Ʒ������������ϡ����ֱ����Ӧֹͣ�����ռ���4.4g������̼��
[�������]
������ѡһ��ͬѧ��ʵ�������������Ǽ������Ʒ��̼���Ƶ������� ��̼���Ƶ����������� ������������ȷ��0.1%��
[������˼]
��1�������С��ͬѧ��Ϊ��Ҫ���̼���Ƶ�������Ҳ����ʹ���������ʯ��ˮ�������ͬ���������� ����һ�־������ʵĻ�ѧʽ������Һ����Ʒ��Ӧ��ͨ���ⶨ������ʵ������������йؼ��㼴�ɣ�
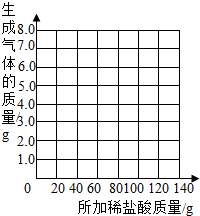
��2�������С��ͬѧ��Ϊ������ϡ�����������������Ҳ�������ȡ13.5g��Ʒ�����ձ��У�ÿ�μ���20gϡ���ᣨ������ˮ���Ȼ����ݳ������þ���������������¼ʵ���������£�
��������a= g��b= g�����������±ߵ�����ֽ�ϻ����������������������ϡ����������ϵ�����ߣ�
[�������]��Ʒ��̼���Ƶ����������Ƕ��٣�
[֪ʶ��]
ʳ�ô������Ҫ�ɷ���̼���ƣ���������������Ȼ��ƣ���Ӧ�����в�����ˮ���Ȼ���Ļӷ���
[��Ʒ�����ʵ��]
����ͬѧ����ȡ12.00��Ʒ����ˮ�����Һ������Һ�м����������ʯ��ˮ�����ˡ�ϴ�ӡ�������õ���ɫ����10.00g��
����ͬѧ����ȡ12.00��Ʒ������������ϡ����ֱ����Ӧֹͣ�����ռ���4.4g������̼��
[�������]
������ѡһ��ͬѧ��ʵ�������������Ǽ������Ʒ��̼���Ƶ�������
[������˼]
��1�������С��ͬѧ��Ϊ��Ҫ���̼���Ƶ�������Ҳ����ʹ���������ʯ��ˮ�������ͬ����������
��2�������С��ͬѧ��Ϊ������ϡ�����������������Ҳ�������ȡ13.5g��Ʒ�����ձ��У�ÿ�μ���20gϡ���ᣨ������ˮ���Ȼ����ݳ������þ���������������¼ʵ���������£�
| ��������Ĵ��� | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
| �ձ�����������������/g | 78.9 | 97.8 | 116.7 | 135.60 | 155.05 | 175.05 | 195.05 |
| �������������/g | 1.1 | 2.2 | a | 4.4 | 4.95 | b | -- |

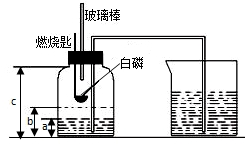 ijУ��ѧ�о���ѧϰС����ѧϰ�ˡ����������������ⶨ���Ļ����ϣ��Ľ��˽̲��е�ʵ�飬��Ƴ�����ͼ��ʾ��ʵ��װ�ã�ʵ�鲽�����£�
ijУ��ѧ�о���ѧϰС����ѧϰ�ˡ����������������ⶨ���Ļ����ϣ��Ľ��˽̲��е�ʵ�飬��Ƴ�����ͼ��ʾ��ʵ��װ�ã�ʵ�鲽�����£�