��Ŀ����
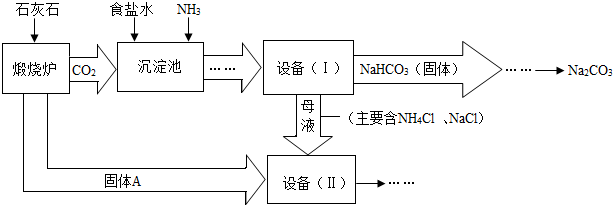
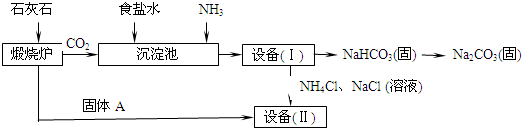
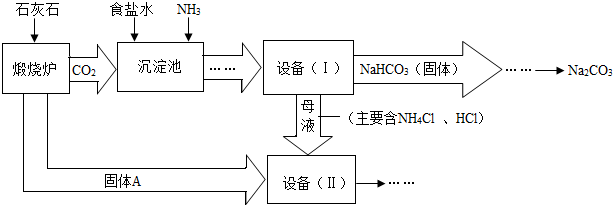
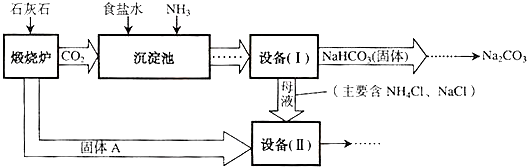
��ҵ�Ͽ�����ʳ�κ�ʯ��ʯΪԭ����ȡ�����ؼ����ڳ���������NaCl��NH3��CO2��H2O�����ܶ��ת����NaHCO3������������NH4Cl����Һ��������Ҫ�����������£�
��֪��2NaHCO3 Na2CO3��CO2����2H2O
Na2CO3��CO2����2H2O
�����������գ�
��1��д���������з�����Ӧ�Ļ�ѧ����ʽ��
��2���ڻ�ѧʵ������Ҫ�����豸�����л�����õ��IJ���������
��3�����豸�����м������Aʱ��������Ӧ�Ļ�ѧ����ʽ�ǣ�
��
��1��NaCl+NH3+CO2+ H2O ="=" NaHCO3�� +NH4Cl (2)����
��3��CaO+ H2O===Ca(OH) 2 Ca(OH) 2+ 2NH4Cl="=" CaCl2+ 2H2O+ 2 NH3��
����

��ϰ��ϵ�д�
�����Ŀ