��Ŀ����
��ѧ��ȤС��ͬѧ�ԡ��������˳�����ݡ������Լ�Ӧ�õȷ���������ܽᣬ�밴Ҫ����գ�
(1)�������˳�����K Ca Na____Sn Pb (H) Cu Hg Ag Pt Au
(2)ͬѧ��ѡ�������������Ƚ�п������ͭ���ֽ����Ļ�ԣ���Zn��Fe��Cu��ϡ�����Fe��ZnSO4��Һ��CuSO4��Һ��Zn��FeSO4��Һ��CuSO4��Һ�����������ʾ��ܴﵽĿ�ĵ���____��������ţ�
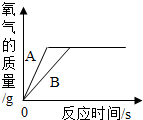
(3)����ͬ����������п��һ������ϡ���ᷴӦ���䷴Ӧ��������ͼ��ʾ��
(1)�������˳�����K Ca Na____Sn Pb (H) Cu Hg Ag Pt Au
(2)ͬѧ��ѡ�������������Ƚ�п������ͭ���ֽ����Ļ�ԣ���Zn��Fe��Cu��ϡ�����Fe��ZnSO4��Һ��CuSO4��Һ��Zn��FeSO4��Һ��CuSO4��Һ�����������ʾ��ܴﵽĿ�ĵ���____��������ţ�
(3)����ͬ����������п��һ������ϡ���ᷴӦ���䷴Ӧ��������ͼ��ʾ��
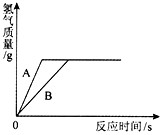
�Իش𣺢�����A��ʾ����____���������п������ϡ���ᷴӦ�������
����������ͼ����˵���ٵĽ����⣬���ɵó��Ľ����ǣ���һ����_________________��
(4)С����ʵ���Һ�л���ͭ���õ���ͭ��Ʒ�к����������ۣ�Ϊ�ⶨ��Ʒ��ͭ�������������������·�������ʵ�飺 ͭ��Ʒ10g�����������ͭ��Һ/��ַ�Ӧ���ˣ�������ϴ�ӡ��������Ϊ10.8g
��д��������Ӧ�ķ���ʽ ____________________�������Ʒ��ͭ����������Ϊ____��
����������ͼ����˵���ٵĽ����⣬���ɵó��Ľ����ǣ���һ����_________________��
(4)С����ʵ���Һ�л���ͭ���õ���ͭ��Ʒ�к����������ۣ�Ϊ�ⶨ��Ʒ��ͭ�������������������·�������ʵ�飺 ͭ��Ʒ10g�����������ͭ��Һ/��ַ�Ӧ���ˣ�������ϴ�ӡ��������Ϊ10.8g
��д��������Ӧ�ķ���ʽ ____________________�������Ʒ��ͭ����������Ϊ____��
(1)Mg Al Zn Fe
(2)�٢�
(3)п����������������һ���������ʣ�ࣨ�������֣�
(4)Fe+CuSO4==FeSO4+Cu��44%
(2)�٢�
(3)п����������������һ���������ʣ�ࣨ�������֣�
(4)Fe+CuSO4==FeSO4+Cu��44%

��ϰ��ϵ�д�
�����Ŀ
 ��ѧ��ȤС��ͬѧ�ԡ��������˳�����ݡ������Լ�Ӧ�õȷ���������ܽᣬ�밴Ҫ����գ�
��ѧ��ȤС��ͬѧ�ԡ��������˳�����ݡ������Լ�Ӧ�õȷ���������ܽᣬ�밴Ҫ����գ�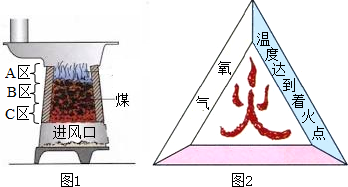

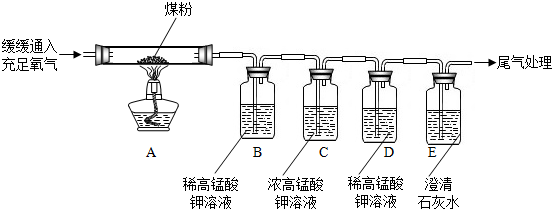
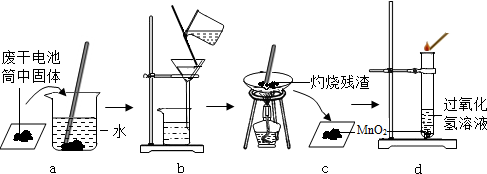
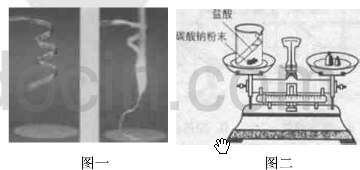 ijУ��ѧ��ȤС��ͬѧ�������غ㶨�ɽ���ʵ��̽��������ͬѧ���ڿ�����ȼ��þ����ʵ����̽��������ͬѧ��ͼ������̽�����ش��������⣺
ijУ��ѧ��ȤС��ͬѧ�������غ㶨�ɽ���ʵ��̽��������ͬѧ���ڿ�����ȼ��þ����ʵ����̽��������ͬѧ��ͼ������̽�����ش��������⣺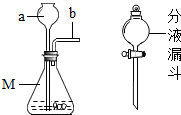 ij��ѧ��ȤС��ͬѧ�ԡ���þ��ҵ����ʹ��CO2�������Ϊ�������ġ����������̽��������������ǵĻ��
ij��ѧ��ȤС��ͬѧ�ԡ���þ��ҵ����ʹ��CO2�������Ϊ�������ġ����������̽��������������ǵĻ��