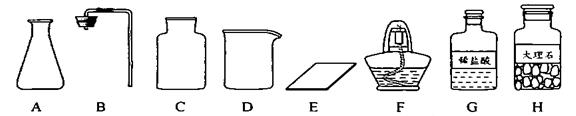
��Ŀ����
�γ���2013���п���ѧʵ�鿼���У�����������ȡ���ڶ�����̼����ȡ��������50g5%��NaCl��Һ����������ǩ����ѧ����ǩȷ��һ��������п��飮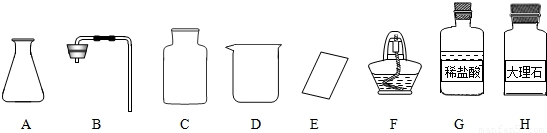
��1����ͬѧ��ǩ������ʵ���ң����ֱ���ʵ��������Ҫ��������������ҩƷ��
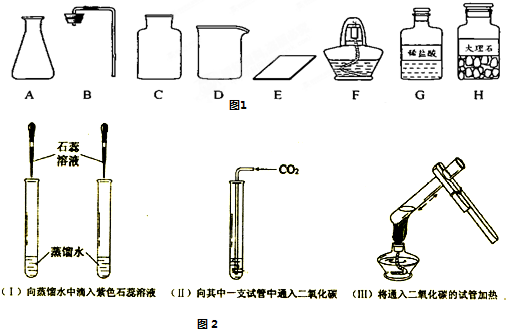
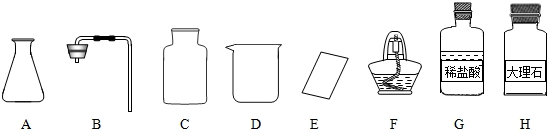
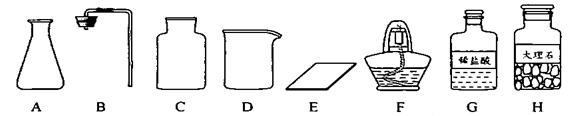
����ͼ������D��������______��F��������______��
�ڼ�ͬѧ�鵽�Ŀ�ǩӦ����______������ĸ���ţ���
A����������ȡ B��������̼����ȡ
����ȡ������ķ�Ӧԭ��Ϊ______���û�ѧ����ʽ��ʾ������ȡһƿ�����壬Ӧѡ�õ�������______������ĸ���ţ���
�ܼ�ͬѧʵ�����Ҫ����ʾ�����£������������������
���װ�á����������ԡ�______���ռ����壮
�������������巢��װ�û�������ȡ�������壬��д������һ�ַ�Ӧ�Ļ�ѧ����ʽ��______ 2H2O+O2��
���𰸡���������1�����������ճ����Ļ�ѧ�������ƺ���;��
�ڸ������������ƺ�ҩƷ����ѡ��
����ȡ������̼��ҩƷ�Ǵ���ʯ��ϡ���ᣬ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼��д����Ӧ�ķ���ʽ�����ݹ����Һ���ڳ����µķ�Ӧѡ����װ�úͶ�����̼���ܶȺ��ܽ���ѡ���ռ���������Ӧ��������
�ܸ���ʵ��IJ��������
�ݸ��ݷ���װ�����ڹ�Һ������װ�ÿ�����ȡ������
ʵ�����ù���������Һ�Ͷ���������ȡ���������������Ǵ�����д����Ӧ�Ļ�ѧ����ʽ��
��2��������Һ�����ʡ��ܼ����������������ļ��㹫ʽ���м��㲢���������������ã�
����⣺��1��������D���������ձ���F�������Ǿƾ��ƣ�
�ڸ��ݸ�����ҩƷϡ����ʹ���ʯ����������ͬѧ�鵽�Ŀ�ǩӦ���ǣ�������̼����ȡ��
����ȡ������̼��ҩƷ��ʯ��ʯ��ϡ���ᣬ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼����Ӧ�ķ���ʽΪ��CaCO3+2HCl=CaCl2+H2O+CO2��������װ���ǹ����Һ���ڳ����µķ�Ӧ����Ҫ�������У�AB����Ϊ������̼������ˮ���ܶȱȿ����ʲ��������ſ������ռ�����Ҫ�������У�CE��
�ܸ÷���ȡ������̼�IJ����ǣ����װ�á����װ�õ������ԡ�װҩƷ���ռ����壻
�ݷ���װ�����ڹ�Һ�����͵�װ�ã���˿�����ʵ�����ù���������Һ�Ͷ���������ȡ���������ж��������Ǵ�������Ӧ�Ļ�ѧ����ʽΪ��2H2O2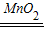 2H2O+O2����
2H2O+O2����
��2������50g5%��NaCl��Һ����Ҫ�Ȼ��Ƶ�����=50g×5%=2.5g����Ҫˮ������=50g-2.5g=47.5g���ܽ�NaClʱ�õ��IJ������������Ǽ����ܽ⣻
�ʴ�Ϊ����1�����ձ����ƾ��ƣ���B����CaCO3+2HCl=CaCl2+H2O+CO2����ABCE��������ҩƷ����2H2O2 2H2O+O2����
2H2O+O2����
��2��2.5��47.5�������ܽ⣮
���������⿼���˳����������ȡװ�á��ռ������ȵ�ѡ���й���Һ�ļ��㣬���ڴ˻����Ͻ�����չ���ؼ�����ȷ����װ�á��ռ�����ѡ������ݣ�
�ڸ������������ƺ�ҩƷ����ѡ��
����ȡ������̼��ҩƷ�Ǵ���ʯ��ϡ���ᣬ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼��д����Ӧ�ķ���ʽ�����ݹ����Һ���ڳ����µķ�Ӧѡ����װ�úͶ�����̼���ܶȺ��ܽ���ѡ���ռ���������Ӧ��������
�ܸ���ʵ��IJ��������
�ݸ��ݷ���װ�����ڹ�Һ������װ�ÿ�����ȡ������
ʵ�����ù���������Һ�Ͷ���������ȡ���������������Ǵ�����д����Ӧ�Ļ�ѧ����ʽ��
��2��������Һ�����ʡ��ܼ����������������ļ��㹫ʽ���м��㲢���������������ã�
����⣺��1��������D���������ձ���F�������Ǿƾ��ƣ�
�ڸ��ݸ�����ҩƷϡ����ʹ���ʯ����������ͬѧ�鵽�Ŀ�ǩӦ���ǣ�������̼����ȡ��
����ȡ������̼��ҩƷ��ʯ��ʯ��ϡ���ᣬ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼����Ӧ�ķ���ʽΪ��CaCO3+2HCl=CaCl2+H2O+CO2��������װ���ǹ����Һ���ڳ����µķ�Ӧ����Ҫ�������У�AB����Ϊ������̼������ˮ���ܶȱȿ����ʲ��������ſ������ռ�����Ҫ�������У�CE��
�ܸ÷���ȡ������̼�IJ����ǣ����װ�á����װ�õ������ԡ�װҩƷ���ռ����壻
�ݷ���װ�����ڹ�Һ�����͵�װ�ã���˿�����ʵ�����ù���������Һ�Ͷ���������ȡ���������ж��������Ǵ�������Ӧ�Ļ�ѧ����ʽΪ��2H2O2
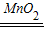 2H2O+O2����
2H2O+O2������2������50g5%��NaCl��Һ����Ҫ�Ȼ��Ƶ�����=50g×5%=2.5g����Ҫˮ������=50g-2.5g=47.5g���ܽ�NaClʱ�õ��IJ������������Ǽ����ܽ⣻
�ʴ�Ϊ����1�����ձ����ƾ��ƣ���B����CaCO3+2HCl=CaCl2+H2O+CO2����ABCE��������ҩƷ����2H2O2
 2H2O+O2����
2H2O+O2������2��2.5��47.5�������ܽ⣮
���������⿼���˳����������ȡװ�á��ռ������ȵ�ѡ���й���Һ�ļ��㣬���ڴ˻����Ͻ�����չ���ؼ�����ȷ����װ�á��ռ�����ѡ������ݣ�

��ϰ��ϵ�д�
�����Ŀ
 ��ͼ������D�������� ��F�������� ��
��ͼ������D�������� ��F�������� ��