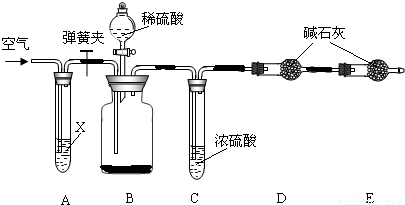
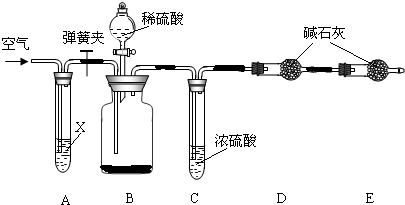
��Ŀ����
ʵ���ҳ��ô��ʯ��ʯ��ˮ��ȡ�������������ƣ�����ȡ�����з�����������ѧ�仯���밴Ҫ��д���������仯�Ļ�ѧ����ʽ��
��1���ֽⷴӦ
��2�����Ϸ�Ӧ
��3�����ֽⷴӦ
��1���ֽⷴӦ
CaCO
CaO+CO2��
| ||
CaCO
CaO+CO2��
��
| ||
��2�����Ϸ�Ӧ
CaO+H2O�TCa��OH��2
CaO+H2O�TCa��OH��2
����3�����ֽⷴӦ
Na2CO3+Ca��OH��2�T2NaOH+CaCO3��
Na2CO3+Ca��OH��2�T2NaOH+CaCO3��
����������д��ѧ����ʽʱ��������Ϥ��Ӧ�������ͷ�Ӧ�������������ݿ���ʵ�����������غ㶨�ɣ�
̼��Ʒֽ����������ƣ���������ˮ���������������ƣ�����������̼���Ʒ�Ӧ�����������ƣ�
̼��Ʒֽ����������ƣ���������ˮ���������������ƣ�����������̼���Ʒ�Ӧ�����������ƣ�
����⣺��1��̼����ڸ��������·ֽ����������ƺͶ�����̼�ǷֽⷴӦ���÷�Ӧ�Ļ�ѧ����ʽΪ��CaCO3
CaO+CO2����
��2����������ˮ��Ӧ�������������ǻ��Ϸ�Ӧ��
�÷�Ӧ�Ļ�ѧ����ʽΪ��CaO+H2O�TCa��OH��2��
��3������������̼���Ʒ�Ӧ�����������ƺ�̼����Ǹ��ֽⷴӦ���÷�Ӧ�Ļ�ѧ����ʽΪ���ʴ�Ϊ��Na2CO3+Ca��OH��2�T2NaOH+CaCO3����
�ʴ�Ϊ��
CaCO3
CaO+CO2����
CaO+H2O�TCa��OH��2��
Na2CO3+Ca��OH��2�T2NaOH+CaCO3����
| ||
��2����������ˮ��Ӧ�������������ǻ��Ϸ�Ӧ��
�÷�Ӧ�Ļ�ѧ����ʽΪ��CaO+H2O�TCa��OH��2��
��3������������̼���Ʒ�Ӧ�����������ƺ�̼����Ǹ��ֽⷴӦ���÷�Ӧ�Ļ�ѧ����ʽΪ���ʴ�Ϊ��Na2CO3+Ca��OH��2�T2NaOH+CaCO3����
�ʴ�Ϊ��
CaCO3
| ||
CaO+H2O�TCa��OH��2��
Na2CO3+Ca��OH��2�T2NaOH+CaCO3����
������������Ҫ���黯ѧ����ʽ����д���ѶȽϴ�

��ϰ��ϵ�д�
�����Ŀ
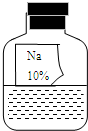 ʵ��������һƿ���õ���ɫҺ���Լ����ܲ�����ɱ�ǩ������ͼ��ʾ�������ȱ�ı�ǩ��ֻʣ�¡�Na���͡�10%��������
ʵ��������һƿ���õ���ɫҺ���Լ����ܲ�����ɱ�ǩ������ͼ��ʾ�������ȱ�ı�ǩ��ֻʣ�¡�Na���͡�10%��������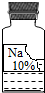 ��2012?Ϣ��һģ��ʵ��������һƿ���õ���ɫ��Һ���ܲ��������ȱ�ı�ǩ��ֻʣ�¡�Na���͡�10%����������ͼ��ʾ����Сǿ��С��ͬѧ�ܸ���Ȥ����������ɷֽ���̽����
��2012?Ϣ��һģ��ʵ��������һƿ���õ���ɫ��Һ���ܲ��������ȱ�ı�ǩ��ֻʣ�¡�Na���͡�10%����������ͼ��ʾ����Сǿ��С��ͬѧ�ܸ���Ȥ����������ɷֽ���̽����