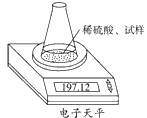
��Ŀ����
��ͼʾװ�òⶨ���Ų��������ֱ��̼���Ƶ��ռ����������Ƶ�������������ȡ��������8.00 g����ƿ����140.00 g����������ϡ����(����Ϊ50.00 g)ÿ����ͬʱ�����һ�Σ��������±���
| �������� | ����/g | |
| ��ƿ+����+ϡ���� | ��1�� | 197.64 |
| ��2�� | 197.48 | |
| ��3�� | 197.26 | |
| ��4�� | 197.16 | |
| ��5�� | 197.12 | |
| ��6�� | 197.12 |
(1)���ؽ��е��ߴζ�����ԭ����___________________________________��
(2)�����������Ƶ�����������
(3)����ϡ������H2SO4���������������ڶ��٣�
������(1)��5�κ͵�6�ζ�����ͬ��˵���ѳ�ַ�Ӧ��
(2)CO2������m(CO2)=(8.00 g+140.00 g+50.00 g)-197.12 g=0.88 g
��������Ʒ��Na2CO3������Ϊx��
Na2CO3+H2SO4====H2O+CO2��+Na2SO4
106 44
x 0.88 g
x=(0.88��106)/44=2.12 g
����ȡ��Ʒ��NaOH����������Ϊ��(5.88/9.00)��100%=73.5%
(3)����Ʒ��Na2CO3����H2SO4������Ϊy��
Na2CO3+H2SO4====H2O+CO2��+Na2SO4
106 98
2.12 g y
y=(2.12 g��98)/106=1.96 g
����������NaOH����H2SO4������Ϊz��
2NaOH+H2SO4====2H2O+Na2SO4
80 98
5.88 z
z=(5.88 g��98)/80=7.20 g
��ϡ������H2SO4����������������
��(1.96 g+7.20 g)/50.00 g�ݡ�100%=18.32%
�𰸣�(1)��5��6�ζ�����ͬ (2)73.5% (3)18.32%

 �����ѧСѧ�꼶�νӽݾ��㽭��ѧ������ϵ�д�
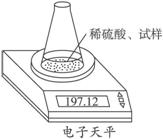
�����ѧСѧ�꼶�νӽݾ��㽭��ѧ������ϵ�д�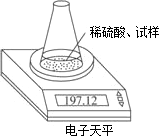 ��ͼʾװ�òⶨ���Ų��������ֱ��̼���Ƶ��ռ����������Ƶ�������������ȡ��������8.00 g����ƿ����140.00 g����������ϡ���ᣨ����Ϊ50.00 g��ÿ����ͬʱ�����һ�Σ��������±���
��ͼʾװ�òⶨ���Ų��������ֱ��̼���Ƶ��ռ����������Ƶ�������������ȡ��������8.00 g����ƿ����140.00 g����������ϡ���ᣨ����Ϊ50.00 g��ÿ����ͬʱ�����һ�Σ��������±���| �������� | ����/g | |
| ��ƿ+����+ϡ���� | ��1�� | 197.64 |
| ��2�� | 197.48 | |
| ��3�� | 197.26 | |
| ��4�� | 197.16 | |
| ��5�� | 197.12 | |
| ��6�� | 197.12 |
��2�������������Ƶ�����������
��3������ϡ������H2SO4���������������ڶ��٣�
��ͼʾװ�òⶨ���Ų��������ֱ��̼���Ƶ��ռ����������Ƶ�������������ȡ��������8.00 g����ƿ����140.00 g����������ϡ����(����Ϊ50.00 g)ÿ����ͬʱ�����һ�Σ��������±���

| �������� | ����/g | |
| ��ƿ+����+ϡ���� | ��1�� | 197.64 |
| ��2�� | 197.48 | |
| ��3�� | 197.26 | |
| ��4�� | 197.16 | |
| ��5�� | 197.12 | |
| ��6�� | 197.12 |
(1)���ؽ��е��ߴζ�����ԭ����___________________________________��
(2)�����������Ƶ�����������
(3)����ϡ������H2SO4���������������ڶ��٣�
��ͼʾװ�òⶨ���Ų��������ֱ��̼���Ƶ��ռ����������Ƶ�������������ȡ��������8.00 g����ƿ����140.00 g����������ϡ����(����Ϊ50.00 g)ÿ����ͬʱ�����һ�Σ��������±���
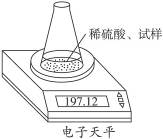
| �������� | ����/g | |
| ��ƿ+����+ϡ���� | ��1�� | 197.64 |
| ��2�� | 197.48 | |
| ��3�� | 197.26 | |
| ��4�� | 197.16 | |
| ��5�� | 197.12 | |
| ��6�� | 197.12 |
(1)���ؽ��е��ߴζ�����ԭ����___________________________________��
(2)�����������Ƶ�����������
(3)����ϡ������H2SO4���������������ڶ��٣�