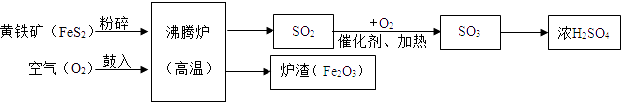
��Ŀ����
����Ŀ��ʵ������һƿδ֪Ũ�ȵ�BaCl2��Һ��ijͬѧȡ��150g����Һ���ձ��У���������μ���������������Ϊ26.5%��Na2CO3��Һ����Ӧ���������ɳ���������������Na2CO3��Һ�����Ĺ�ϵ��ͼ��ʾ������㣺 
��1������26.5%��Na2CO3��Һ80g����ҪNa2CO3����g��
��2��BaCl2��Һ���������������Ƕ��٣���д��������̣����������0.1%��
��3����Ӧǰ����Һ�б�Ԫ�ص����������������С�����䡱����
���𰸡�
��1��21.2
��2���⣺��BaCl2��Һ����������Ϊx
BaCl2+ | Na2CO3=BaCO3��+2NaCl |
208 | 106 |
x | 10.6g |
![]() =
= ![]()
x=20.8g
BaCl2��Һ��������������Ϊ ![]() ��100%��13.9%
��100%��13.9%
���ڷ�Ӧ�����б�Ԫ��������״̬ת��Ϊ����״̬��������Һ�еı�Ԫ�ص������ڼ��٣�
�𣺷�Ӧǰ����Һ�б�Ԫ�ص����� ��С
��3����С
���������⣺����26.5%��Na2CO3��Һ80g����ҪNa2CO3���������Ϊ26.5%��80g=21.2g
��ͼ��֪����40g��������Ϊ26.5%��̼������Һʱ�������ﵽ���������ʱ�μӷ�Ӧ��̼���Ƶ�����Ϊ26.5%��40g=10.6g��1������26.5%��Na2CO3��Һ80g����ҪNa2CO3���� 21.2g����2��BaCl2��Һ��������������Ϊ13.9%��
�����㾫����������Ĺؼ�����������ݻ�ѧ��Ӧ����ʽ�ļ�������֪ʶ�����ո����ʼ�������=ϵ������Է�������֮�ȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ����ѧ���Ͻ�ʦ��ʾ�˼���̿��(����)������ͭ������ʵ�飬�κ����С��ͬѧ�Խ�ʦ��ʾ�л�õĻҺ�ɫ��ĩ��Ʒ����ʵ�顣ʵ��װ������ͼ��ʾ��ͼ������̨��װ������ȥ��D��ʢ�ŵ��ǹ����������ƺ������ƵĻ�����
ʵ����Ҫԭ������ȡ��Ʒ��ĩW g������C�У�D��װ��agҩƷ����Ӧ��������ȴ����D��ҩƷ����Ϊb g����Ʒ��ͭ������������[w��(b��a)��12/44]/w
��ش��������⡣
����Ҫʵ������
ʵ����Ҫ���� | ʵ������ | �йػ�ѧ����ʽ |
��A�������������������μ���Һ | B��E�������� | |
��C���м��ȣ���C��ҩƷ��ַ�ӳ�ر�A�Ļ�����ֹͣ���� | C+O2 |
��ʵ������
װ��E��ʢ�ŵ��Լ��� �� ����������
������ʵ�飬�õ�ͭ��������������ȷ������Ϊ�����ƫ��ƫС������ȷ������ԭ���� �� ���Ҫ�õ��˽���ȷ�Ľ�����Ը�װ�øĽ��ķ�������