��Ŀ����
ijУ������ȤС���һ����п��Ʒ���������ʣ����ʲ�����ˮ��Ҳ�����ᷴӦ�����з������ס��ҡ�������ͬѧ�ֱ����ʵ�飬ʵ�����ݼ�¼���£�
| ҩƷ ��� | �� | �� | �� |
| �ձ�+ϡ����/g | 152.5 | 152.5 | 252.5 |
| ��п��Ʒ | 20 | 15 | 15 |
| �ձ���ʣ����/g | 172.1 | 167.1 | 267.1 |
��������������ݣ��������ǻش��������⣨ÿ�鷴Ӧ����֣���
��1��______��ͬѧ��ȡ�õ�ϡ�������п��Ʒǡ����ȫ��Ӧ��
��2�������п��Ʒ��п����������______��
��3������ԭ������Һ�����ʵ���������______��
��4������ǡ����ȫ��Ӧ������ʵ��������Һ�����ʵ������������ձ�����Ϊ52.5g����������ȷ��0.1%��______��
���������غ㶨�ɣ�����H2������=152.5g+15g-167.1g=0.4g
����Ʒ��п������Ϊx����Ӧ����H2SO4������Ϊy������ZnSO4������Ϊz��
Zn+H2SO4�TZnSO4+H2��
65 98 161 2
x y z 0.4g
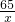 =
=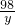 =
=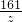 =
=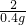
��֮�ã�x=13g��y=19.6g��z=32.2g
��2��п��Ʒ��п����������=
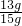 ��100%=86.7%
��100%=86.7%��3��ԭ������Һ�����ʵ���������=
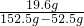 ��100%=19.6%
��100%=19.6%��4��������Һ�����ʵ���������=
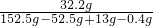 ��100%=28.6%
��100%=28.6%�ʴ�Ϊ����1���ң���2��86.7%����3��19.6%����4��28.6%��
�������Աȼס��ҡ�������ʵ���е�ʵ�����ݣ����鶼�õ�0.4g���������жϵõ�����ʵ����ȡ�õ�ϡ�������п��Ʒǡ����ȫ��Ӧ�����÷�Ӧ�ų����������������ݷ�Ӧ�Ļ�ѧ����ʽ���������Ʒ��п����������Ӧ�����������������Ӧ����������п�����������������ؼ��㣮
���������ݷ�Ӧ�Ļ�ѧ����ʽ���Ա�ʾ��Ӧ�и����ʵ������ȣ��ɷ�Ӧ��ijһ���ʵ������ɼ��㷴Ӧ���������ʵ�������

 ��Կ���Ծ�ϵ�д�
��Կ���Ծ�ϵ�д�ijУ��ѧ��ȤС���ڿ����У���ij��Һ�����˶�μ��飬����3�μ���Ľ�����±���ʾ��
| ��� | ��Һ�м���������� |
| 1 | KCl��K2SO4��Na2CO3 |
| 2 | Na2SO4��BaCl2��K2CO3 |
| 3 | Na2SO4��KCl��K2CO3 |
�Ķ��ϱ���ش�
��1�����е�________�ν���϶���ȷ��
��2��Ϊ��֤����2�μ��������Ƿ����SO42����CO32����Cl�����ֽ������²������ټ��������___________�����������ƣ���ͬ����Ŀ���Ǽ���__________�������ȥ���ڼ���_________��Һ�����ˣ�������Һ�м���__________��Һ����С��ͬѧ�������Ϻ��ֿ���������ɫ��Ӧ��ȷ����Һ�д��ڵ������ӡ�
��3��ͨ������ʵ�飬��С�黹�Ǿ�����ȷ������2�μ���������һ������ȷ�ģ�����Ϊ���ܵ�ԭ����_________________________________________________________________��
| ��� | ��Һ�м���������� |
| 1 | KCl��K2SO4��Na2CO3 |
| 2 | Na2SO4��BaCl2��K2CO3 |
| 3 | Na2SO4��KCl��K2CO3 |
��1�����е� �ν���϶���ȷ��
��2��Ϊ��֤����2�μ��������Ƿ����SO42-��CO32-��Cl-���ֽ������²������ټ�������� �����������ƣ���ͬ����Ŀ���Ǽ��� �������ȥ���ڼ��� ��Һ�����ˣ�������Һ�м��� ��Һ����С��ͬѧ�������Ϻ��ֿ���������ɫ��Ӧ��ȷ����Һ�д��ڵ������ӣ�
��3��ͨ������ʵ�飬��С�黹�Ǿ�����ȷ������2�μ���������һ������ȷ�ģ�����Ϊ���ܵ�ԭ���� ��
| ��� | ��Һ�м���������� |
| 1 | KCl��K2SO4��Na2CO3 |
| 2 | Na2SO4��BaCl2��K2CO3 |
| 3 | Na2SO4��KCl��K2CO3 |
��1�����е� �ν���϶���ȷ��
��2��Ϊ��֤����2�μ��������Ƿ����SO42-��CO32-��Cl-���ֽ������²������ټ�������� �����������ƣ���ͬ����Ŀ���Ǽ��� �������ȥ���ڼ��� ��Һ�����ˣ�������Һ�м��� ��Һ����С��ͬѧ�������Ϻ��ֿ���������ɫ��Ӧ��ȷ����Һ�д��ڵ������ӣ�
��3��ͨ������ʵ�飬��С�黹�Ǿ�����ȷ������2�μ���������һ������ȷ�ģ�����Ϊ���ܵ�ԭ���� ��