��Ŀ����
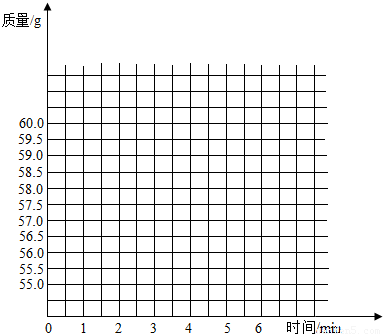
��ʢ��50gϡ������ձ�������ƽ�ϣ�Ȼ��ѹ�����̼���Ƽ��뵽ʢ��ϡ������ձ��У��Բⶨϡ������HCl��������������֪��Ӧǰ�ձ���ϡ�����̼���Ƶ�������Ϊ60.0g���ӷ�Ӧ��ʼ���Ժ��6�����ڣ�ÿ1���Ӷ�1��������¼��������£�| ʱ��/min | 1 | 2 | 3 | 4 | 5 | 6 | |
| ����/g | 60.0 | 58.0 | 57.0 | 56.8 | 56.7 | 57.2 | 56.7 |
��1��������һ���ⶨ������ϴ�������ڵ�ʱ���ǵ�______min��
��2���ڸ���������ֽ�ϣ�������ʾ��Ӧ���̵�������ʱ�������ͼ
 ��
����3���Լ���50gϡ������HCl������������д��������̣���
���𰸡���������1������̼������ϡ���ᷴӦʱ���ձ������ʵ�������Ӧ����С����ֲ��䣮���ڵ�5����ʱ�ձ�����������������˵���ⶨ������ϴ�
��2�����ݱ��������ȵ����Ӧ�ĵ㣬Ȼ���������߽��������������ɣ�
��3���ɼ��ٵ���������̼���������ᷴӦ�Ļ�ѧ����ʽ���Լ����ϡ���������ʵ�����������
����⣺��1������̼������ϡ���ᷴӦʱ���ձ������ʵ�������Ӧ����С����ֲ��䣮���ڵ�5����ʱ�ձ�����������������˵���ⶨ������ϴ�
��2����ͼ
 ��
��
��3����ϡ���������ʵ���������Ϊx��
Na2CO3+2HCl=2NaCl+H2O+CO2��
73 44
50g×x 60.0g-56.7g
 =
= ��x=10.95%
��x=10.95%
�𣺣�3��ϡ���������ʵ���������Ϊ10.95%��
��������1����Ӧ�������ձ��е�������������ֻ�ܼ��٣��������ӣ��ɴ˿����ҳ��������ϴ�ķ�Χ��
��2�����ߵĻ���Ӧ�����ѧ�����ߵĻ�����
��3���ɼ��ٵ���������̼���������ᷴӦ�Ļ�ѧ����ʽֱ�Ӽ��������ϡ���������ʵ�������������ϡ���������ʵ�����������
��2�����ݱ��������ȵ����Ӧ�ĵ㣬Ȼ���������߽��������������ɣ�
��3���ɼ��ٵ���������̼���������ᷴӦ�Ļ�ѧ����ʽ���Լ����ϡ���������ʵ�����������
����⣺��1������̼������ϡ���ᷴӦʱ���ձ������ʵ�������Ӧ����С����ֲ��䣮���ڵ�5����ʱ�ձ�����������������˵���ⶨ������ϴ�
��2����ͼ
 ��
����3����ϡ���������ʵ���������Ϊx��
Na2CO3+2HCl=2NaCl+H2O+CO2��
73 44
50g×x 60.0g-56.7g
 =
= ��x=10.95%
��x=10.95%�𣺣�3��ϡ���������ʵ���������Ϊ10.95%��
��������1����Ӧ�������ձ��е�������������ֻ�ܼ��٣��������ӣ��ɴ˿����ҳ��������ϴ�ķ�Χ��
��2�����ߵĻ���Ӧ�����ѧ�����ߵĻ�����
��3���ɼ��ٵ���������̼���������ᷴӦ�Ļ�ѧ����ʽֱ�Ӽ��������ϡ���������ʵ�������������ϡ���������ʵ�����������

��ϰ��ϵ�д�
�����Ŀ
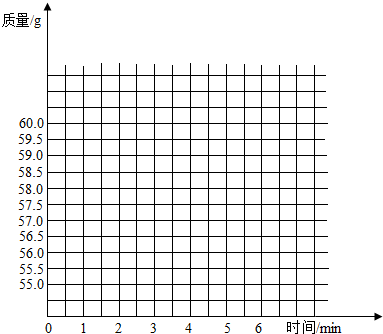
��ʢ��50gϡ������ձ�������ƽ�ϣ�Ȼ��ѹ�����̼���Ƽ��뵽ʢ��ϡ������ձ��У��Բⶨϡ������HCl��������������֪��Ӧǰ�ձ���ϡ�����̼���Ƶ�������Ϊ60.0g���ӷ�Ӧ��ʼ���Ժ��6�����ڣ�ÿ1���Ӷ�1��������¼��������£�
�Իش��������⣺
��1��������һ���ⶨ������ϴ�������ڵ�ʱ���ǵ� min��
��2���ڸ���������ֽ�ϣ�������ʾ��Ӧ���̵�������ʱ�������ͼ
 ��
��
��3���Լ���50gϡ������HCl������������д��������̣���
| ʱ��/min | 0 | 1 | 2 | 3 | 4 | 5 | 6 |
| ����/g | 60.0 | 58.0 | 57.0 | 56.8 | 56.7 | 57.2 | 56.7 |
��1��������һ���ⶨ������ϴ�������ڵ�ʱ���ǵ�
��2���ڸ���������ֽ�ϣ�������ʾ��Ӧ���̵�������ʱ�������ͼ
 ��
����3���Լ���50gϡ������HCl������������д��������̣���
��ʢ��50gϡ������ձ�������ƽ�ϣ�Ȼ��ѹ�����̼���Ƽ��뵽ʢ��ϡ������ձ��У��Բⶨϡ������HCl��������������֪��Ӧǰ�ձ���ϡ�����̼���Ƶ�������Ϊ60.0g, �ӷ�Ӧ��ʼ���Ժ��6�����ڣ�ÿ1���Ӷ�1��������¼��������£�

�Իش��������⣺
��1��������һ���ⶨ������ϴ�������ڵ�ʱ���ǵ�_______min��
��2���ڸ���������ֽ�ϣ�������ʾ��Ӧ���̵�������ʱ�������ͼ��
��3���Լ���50gϡ������HCl������������д��������̣���

| ʱ��/min | 0 | 1 | 2 | 3 | 4 | 5 | 6 |
| ����/g | 60.0 | 58.0 | 57.0 | 56.8 | 56.7 | 57.2 | 56.7 |
��1��������һ���ⶨ������ϴ�������ڵ�ʱ���ǵ�_______min��
��2���ڸ���������ֽ�ϣ�������ʾ��Ӧ���̵�������ʱ�������ͼ��
��3���Լ���50gϡ������HCl������������д��������̣���
��ʢ��50gϡ������ձ�������ƽ�ϣ�Ȼ��ѹ�����̼���Ƽ��뵽ʢ��ϡ������ձ��У��Բⶨϡ������HCl��������������֪��Ӧǰ�ձ���ϡ�����̼���Ƶ�������Ϊ60.0g, �ӷ�Ӧ��ʼ���Ժ��6�����ڣ�ÿ1���Ӷ�1��������¼��������£�
| ʱ��/min | 0 | 1 | 2 | 3 | 4 | 5 | 6 |
| ����/g | 60.0 | 58.0 | 57.0 | 56.8 | 56.7 | 57.2 | 56.7 |
��1��������һ���ⶨ������ϴ�������ڵ�ʱ���ǵ�_______min��
��2���ڸ���������ֽ�ϣ�������ʾ��Ӧ���̵�������ʱ�������ͼ��
��3���Լ���50gϡ������HCl������������д��������̣���
��ʢ��50gϡ������ձ�������ƽ�ϣ�Ȼ��ѹ�����̼���Ƽ��뵽ʢ��ϡ������ձ��У��Բⶨϡ������HCl��������������֪��Ӧǰ�ձ���ϡ�����̼���Ƶ�������Ϊ60.0g���ӷ�Ӧ��ʼ���Ժ��6�����ڣ�ÿ1���Ӷ�1��������¼��������£�
| ʱ��/min | 0 | 1 | 2 | 3 | 4 | 5 | 6 |
| ����/g | 60.0 | 58.0 | 57.0 | 56.8 | 56.7 | 57.2 | 56.7 |
��1��������һ���ⶨ������ϴ�������ڵ�ʱ���ǵ�________min��
��2���ڸ���������ֽ�ϣ�������ʾ��Ӧ���̵�������ʱ�������ͼ
 ��
����3���Լ���50gϡ������HCl������������д��������̣���