��Ŀ����
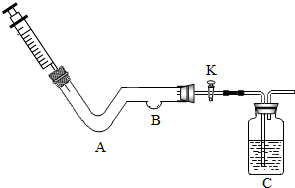 ij��ѧС��ͬѧ������ͼ��ʾװ�ý���ʵ�飮
ij��ѧС��ͬѧ������ͼ��ʾװ�ý���ʵ�飮��1��A����ʯ��ʯ��飬B��������ʯ����Һ����ע����ע������ϡ���ᣬͬʱ��K��һ��ʱ��۲쵽B����������
��2��A���Ŷ������̣�B����ľ̿��C�зų���ʯ��ˮ��Ϊ��֤��ȼ��ȼ�յ���������������������ʵ�飺
�ٴ�K����ע����ע��һ����������
��һ��ʱ�����ע����ע�������Ĺ���������Һ��ͬʱ��K��A���������壬ľ̿��ȼ�գ�����ʯ��ˮ
���㣺������̼��ʵ�����Ʒ�,ʵ������ȡ�����ķ�Ӧԭ��,̼�Ļ�ѧ����,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ,ȼ����ȼ�յ�����
ר�⣺���������ʵ�����Ʒ������顢�����뾻��,��ѧ̽��
��������1����ʯ��ʯ��ϡ���ᷴӦ���ɶ�����̼��������̼��ˮ��Ӧ����̼�ᣬ̼�������ᣬ�ܹ�ʹʯ����Һ���ȥ�������
��2����û��������ľ̿����ȼ�գ�ͬ���Ǻ������Ӵ������ȴﵽ�Ż���ȼ�գ������ȾͲ�ȼ�գ�˵��ȼ����Ҫ�ﵽ��ȼ����Ż��ȥ�������
��2����û��������ľ̿����ȼ�գ�ͬ���Ǻ������Ӵ������ȴﵽ�Ż���ȼ�գ������ȾͲ�ȼ�գ�˵��ȼ����Ҫ�ﵽ��ȼ����Ż��ȥ�������
����⣺��1��ʯ��ʯ��ϡ���ᷴӦ���ɶ�����̼��������̼��ˮ��Ӧ����̼�ᣬ�仯ѧ����ʽΪ��H2O+CO2�TH2CO3��̼�������ᣬ�ܹ�ʹʯ����Һ��죻�ʴ�Ϊ����ɫʯ����Һ��� H2O+CO2�TH2CO3��
��2������ע����ע��һ��������������B����ʹ�¶ȴﵽľ̿���Ż�㣬����û��������ľ̿����ȼ�գ��ɴ�֤������ȼ��ʱ��Ҫ�������ʴ�Ϊ������B�� ����ȼ����Ҫ������
�ڹ���������Һ�Ͷ������̷�Ӧ��������������ľ̿û�дﵽ�Ż�㣬����ľ̿����ȼ�գ�Ҳ�Ͳ������������̼������ʯ��ˮ������ǣ�˵������ֻ����������ȼ�գ���B����ȼ�ƾ��ƣ����ﵽľ̿���Ż����������Ӵ���ľ̿ȼ�գ�ͬ���Ǻ������Ӵ������ȴﵽ�Ż���ȼ�գ������ȾͲ�ȼ�գ�˵��ȼ����Ҫ�ﵽ��ȼ����Ż�� �������ɵĶ�����̼�ͳ���ʯ��ˮ��Ӧ���ɲ�����ˮ��̼��ƺ�ˮ���䷴Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O���ʴ�Ϊ��������� CO2+Ca��OH��2�TCaCO3��+H2O ȼ����Ҫ�ﵽ��ȼ����Ż�㣮
��2������ע����ע��һ��������������B����ʹ�¶ȴﵽľ̿���Ż�㣬����û��������ľ̿����ȼ�գ��ɴ�֤������ȼ��ʱ��Ҫ�������ʴ�Ϊ������B�� ����ȼ����Ҫ������
�ڹ���������Һ�Ͷ������̷�Ӧ��������������ľ̿û�дﵽ�Ż�㣬����ľ̿����ȼ�գ�Ҳ�Ͳ������������̼������ʯ��ˮ������ǣ�˵������ֻ����������ȼ�գ���B����ȼ�ƾ��ƣ����ﵽľ̿���Ż����������Ӵ���ľ̿ȼ�գ�ͬ���Ǻ������Ӵ������ȴﵽ�Ż���ȼ�գ������ȾͲ�ȼ�գ�˵��ȼ����Ҫ�ﵽ��ȼ����Ż�� �������ɵĶ�����̼�ͳ���ʯ��ˮ��Ӧ���ɲ�����ˮ��̼��ƺ�ˮ���䷴Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O���ʴ�Ϊ��������� CO2+Ca��OH��2�TCaCO3��+H2O ȼ����Ҫ�ﵽ��ȼ����Ż�㣮
���������������˶�����̼�����ʼ���ѧ����ʽ����д��ȼ�յ��������ǿ��Ժ��ص㣮

��ϰ��ϵ�д�
 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�
�����Ŀ
 2013��12��2��1ʱ30�֣��ҹ��ɹ������϶����š�̽�����������գ������úš����Ľṹ������Ϊ�ҹ��������Ƶ�����þ�Ͻ���ϣ�����þ�Ͻ����ڣ�������
2013��12��2��1ʱ30�֣��ҹ��ɹ������϶����š�̽�����������գ������úš����Ľṹ������Ϊ�ҹ��������Ƶ�����þ�Ͻ���ϣ�����þ�Ͻ����ڣ�������| A������� | B�������� |
| C������ | D�������� |
���б�ʾ���ij��ϡ�������ԭ�ӽṹʾ��ͼ�ǣ�������
A��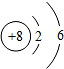 |
B�� |
C��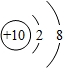 |
D��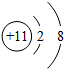 |
����ʵ�������װ����ȷ���ǣ�������
A��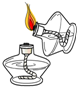 ��ȼ�ƾ��� |
B��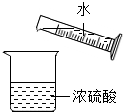 ϡ��Ũ���� |
C��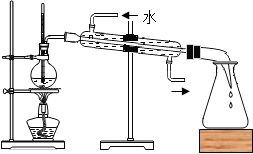 ���� |
D��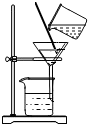 ���� |
 ��֪A��G��������Ϥ�����ʣ�����A��E��F��G���ǵ��ʣ�B��C��D���������A��B��C���ǿ����ijɷ֣�E�Ǻ�ɫ���壬F��Ŀǰ������ߵĽ���������֮������ͼ��ʾ�ķ�Ӧ��ת����ϵ��ͼ�С�-����ʾ���������ܷ�Ӧ����������ʾת����ϵ����
��֪A��G��������Ϥ�����ʣ�����A��E��F��G���ǵ��ʣ�B��C��D���������A��B��C���ǿ����ijɷ֣�E�Ǻ�ɫ���壬F��Ŀǰ������ߵĽ���������֮������ͼ��ʾ�ķ�Ӧ��ת����ϵ��ͼ�С�-����ʾ���������ܷ�Ӧ����������ʾת����ϵ���� �������������������������Ź㷺��Ӧ�ã�
�������������������������Ź㷺��Ӧ�ã�