��Ŀ����
ijƷ�ƴ����Ʒ�к��������Ȼ������ʣ����Ʒ��װ���ϱ�����̼���ơ�96%��Ϊ�ⶨ�ò�Ʒ�к�̼���Ƶ���������������������ʵ�飻ȡ11.0g������Ʒ�����ձ��У��Ƶ��ձ�����ʢ������Ʒ��������Ϊ158.0g���ٰ�100gϡ����ƽ���ֳ��ķ����μ�����Ʒ�У���ַ�Ӧ��ʵ�����ݼ�¼���£�
����ݴ˷������㣺
��1����һ�μ���ϡ�����ַ�Ӧ�����ɶ�����̼��������______ g��
��2���ò�Ʒ��̼���Ƶ�����������
| ��������Ĵ��� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
| �ձ�����ʢ���ʵ�������/g | 181.2 | 204.4 | 228.6 | 253.6 |
��1����һ�μ���ϡ�����ַ�Ӧ�����ɶ�����̼��������______ g��
��2���ò�Ʒ��̼���Ƶ�����������
��1���ɱ������ݿ�֪����һ�����ɶ�����̼������Ϊ��158.0g+100g��4-181.2g=1.8g�����1.8��
��2����ַ�Ӧ�����ɶ�����̼������Ϊ��158.0g+100g-253.6g=4.4g
����Ʒ�к��е�̼���Ƶ�����Ϊx
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 44
x 4.4g
=
x=10.6g
̼���Ƶ���������Ϊ��
��100%=96.4%
�𣺸ò�Ʒ��̼���Ƶ���������Ϊ96.4%��
��2����ַ�Ӧ�����ɶ�����̼������Ϊ��158.0g+100g-253.6g=4.4g
����Ʒ�к��е�̼���Ƶ�����Ϊx
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 44
x 4.4g
| 106 |
| x |
| 44 |
| 4.4g |
x=10.6g
̼���Ƶ���������Ϊ��
| 10.6g |
| 11.0g |
�𣺸ò�Ʒ��̼���Ƶ���������Ϊ96.4%��

��ϰ��ϵ�д�
�����Ŀ
ijƷ�ƴ����Ʒ�к��������Ȼ������ʣ����Ʒ��װ���ϱ�����̼���ơ�96%��Ϊ�ⶨ�ò�Ʒ�к�̼���Ƶ���������������������ʵ�飻ȡ11.0g������Ʒ�����ձ��У��Ƶ��ձ�����ʢ������Ʒ��������Ϊ158.0g���ٰ�100gϡ����ƽ���ֳ��ķ����μ�����Ʒ�У���ַ�Ӧ��ʵ�����ݼ�¼���£�
| ��������Ĵ��� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
| �ձ�����ʢ���ʵ�������/g | 181.2 | 204.4 | 228.6 | 253.6 |
��1����һ�μ���ϡ�����ַ�Ӧ�����ɶ�����̼��������______ g��
��2���ò�Ʒ��̼���Ƶ�����������
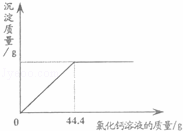
 ��2013?�ൺ���������Ĵ����Ʒ�����������������Ȼ��ƣ�ij��ѧ��ȤС���ͬѧͨ��ʵ�����ⶨijƷ�ƵĴ�����Ʒ��̼���Ƶ���������������ȡ��5g�ô�����Ʒ�����ձ������Ƴ�60g��Һ������������μ���10%���Ȼ�����Һ����ü����Ȼ��Ƶ����������������������ϵ��ͼ��ʾ����ش��������⣺
��2013?�ൺ���������Ĵ����Ʒ�����������������Ȼ��ƣ�ij��ѧ��ȤС���ͬѧͨ��ʵ�����ⶨijƷ�ƵĴ�����Ʒ��̼���Ƶ���������������ȡ��5g�ô�����Ʒ�����ձ������Ƴ�60g��Һ������������μ���10%���Ȼ�����Һ����ü����Ȼ��Ƶ����������������������ϵ��ͼ��ʾ����ش��������⣺