��Ŀ����
�ӹŴ����ִ������ϵ�����Խ��Խ�࣬��;ҲԽ��Խ�㷺�� ��ش�
(1)��������ʱ�ڣ��ҹ�����ʪ����ͭ�ļ��أ�������������ͭ��Һ�еõ�ͭ���ʣ��÷�Ӧ����_______________(�������Ӧ����)��
(2)�ִ���ᣬ������һ�ֳ����ķǽ������ϣ�SiO2������������ԭ��֮һ��SiO2����___________ (ѡ��ᡱ���� ����� �Ρ��������)��
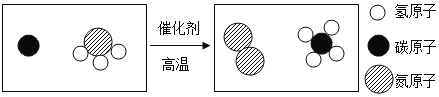
(3)δ������������������ʱ���ע�ػ�����������������̼��Ϊԭ��Ӧ���ڡ���ɫ����ѧ���Ը��õر�������������ÿ������Դ���Ѷ�������ŷ�Լ200�ڶֵ�CO2������Щ CO2����ѧ����ʽ��CO2+3H2 CH3OH+H2O��"���з�Ӧ�������Ͽ���ת��Ϊ_______�ڶּ״�[CH3OH](�������һλС��)��
CH3OH+H2O��"���з�Ӧ�������Ͽ���ת��Ϊ_______�ڶּ״�[CH3OH](�������һλС��)��
�����ࡱ��ѧϰ��ѧ����Ҫ����֮һ�����з�����ȷ���ǣ�������
ѡ�� ��� | ����� | ���� | ������ | �� |
A | �Ҵ����� | ���ʯ | CaO | ���� |
B | ú | ���� | �ɱ� | H3PO4 |
C | Ӳˮ | �� | Fe2O3 | NH4HCO3 |
D | ���� | He | CH4O | ���� |
A. A B. B C. C D. D
Сά����˾۲�ʱ���Ի��ȼ�ϡ�����ƾ��������˺��棬��������ͬѧ����ɷֽ�������̽����
���������ϣ���1���ù���ƾ����þƾ����Ȼ��ƺ��������ư�һ���������Ȼ���Ƴɵġ�
��2���Ȼ��ơ��Ȼ�����Һ�������ԡ�
��������⣩��1���ƾ����Ƿ���̼Ԫ�أ� ��2������ƾ������������Ƿ���ʣ�

��ʵ��̽������1����ͼ��ʾ����ʵ�飬���ֳ���ʯ��ˮ_____,�ɵó��ƾ��к���̼Ԫ�صĽ��ۡ�
��2��ȡ��������ƾ����ձ��У���������ˮ����ܽ���ã������ձ��ײ��а�ɫ���������û�ѧ����ʽ��ʾ�ó���������γɵģ�_____���ɴ�˵�����������ѱ��ʡ�
��ʵ����ۣ���1���ƾ��к���̼Ԫ�ء� ��2������ƾ����������Ƽ������ʡ�
����չ���죩Ϊ��һ��ȷ���������Ƶı��ʳ̶ȣ��������̽��������ͬѧȡ�ձ��ϲ���Һ����֧�Թ��У�����ͼ��ʾ����ʵ�顣
ʵ�鷽�� |
|
|
ʵ������ | ��Һ_____ | ������ɫ���� |
ʵ����� | ��Һ������������ | ��Һ����____ |
����ͬѧ��Ϊ����ʵ�鲻��֤����Һ��һ�����������ƣ�������_____��������ȡ�ձ��е��ϲ���Һ���������Ȼ�����Һ����ַ�Ӧ���ã�ȡ�ϲ���Һ���μӷ�̪��Һ����̪��Һ��죬֤����Һ��һ�����������ơ�
����˼����������ʵ���м������Ȼ�����Һ��Ŀ����_____��
��ʵ����ۣ�С��ͬѧ�������ۣ�һ����Ϊ�ù���ƾ��е��������Ʋ��ֱ��ʡ�