��Ŀ����
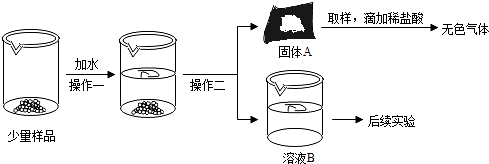
 ijС��ͬѧ����ͼ��ʾ��ԭ������������ش��������⣮
ijС��ͬѧ����ͼ��ʾ��ԭ������������ش��������⣮��1��ʵ����ʼʱ��Ӧ
��2����
��3���÷�Ӧ�Ļ�ѧ����ʽΪ
��4��ʵ������й۲쵽����ʯ��ˮ�����ǣ�˵������
��5���ұߵ����ܿڷ��õ�ȼ�ľƾ��Ƶ�Ŀ����
���㣺һ����̼��ԭ������,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺�������������
��������1����ȼ�Ե�����Ϳ��������������Ļ������ ��ȼʱ�ᷢ����ը��
��2�����ݷ�Ӧ������
��3�����ݷ�Ӧ���������Լ���Ӧ����ȷ������ʽ��
��4��������̼��ʹ�����ʯ��ˮ����ǽ��
��5������һ����̼�ж�������Ⱦ����������
��2�����ݷ�Ӧ������
��3�����ݷ�Ӧ���������Լ���Ӧ����ȷ������ʽ��
��4��������̼��ʹ�����ʯ��ˮ����ǽ��
��5������һ����̼�ж�������Ⱦ����������
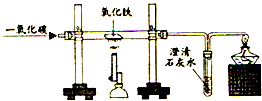
����⣺��1��һ����̼���п�ȼ�Ժ���������ڵ�ȼ�������»ᷢ����ը�������ԣ���ʼʱ��Ӧ��ͨһ��CO��Ŀ���ǣ��ž�װ���ڵĿ�������ֹ����ʱ������ը��
��2��һ����̼��ԭ�������ڼ��ȵ��������������Ͷ�����̼���������Ǻ�ɫ��ĩ�����Ǻ�ɫ�ķ�ĩ�����ԣ�ʵ��������ǣ�����ɫ������ɺ�ɫ��
��3����Ӧ�ķ���ʽ�ǣ�3CO+Fe2O3
2Fe+3CO2��
��4�����ڶ�����̼��ʹ�����ʯ��ˮ����ǣ����ԣ�ʵ��װ���г���ʯ��ˮ�����˵���ж�����̼���ɣ���ѧ����ʽΪ��CO2+Ca��OH��2=CaCO3��+H2O��
��5������β���к���һ����̼��һ����̼�ж�������Ⱦ���������ԣ��ұߵ����ܿڷ��õ�ȼ�ľƾ�������ȥʣ���һ����̼��
�ʴ�Ϊ����1����ͨCO�ټ��ȣ��ž�װ���ڿ����Է�ֹ��ը����2����ɫ��ĩ����ɫ��ĩ����3��3CO+Fe2O3
2Fe+3CO2��
��4��������̼��CO2+Ca��OH��2=CaCO3��+H2O����5����ֹδ��Ӧ��һ����̼��Ⱦ������
��2��һ����̼��ԭ�������ڼ��ȵ��������������Ͷ�����̼���������Ǻ�ɫ��ĩ�����Ǻ�ɫ�ķ�ĩ�����ԣ�ʵ��������ǣ�����ɫ������ɺ�ɫ��
��3����Ӧ�ķ���ʽ�ǣ�3CO+Fe2O3
| ||
��4�����ڶ�����̼��ʹ�����ʯ��ˮ����ǣ����ԣ�ʵ��װ���г���ʯ��ˮ�����˵���ж�����̼���ɣ���ѧ����ʽΪ��CO2+Ca��OH��2=CaCO3��+H2O��
��5������β���к���һ����̼��һ����̼�ж�������Ⱦ���������ԣ��ұߵ����ܿڷ��õ�ȼ�ľƾ�������ȥʣ���һ����̼��
�ʴ�Ϊ����1����ͨCO�ټ��ȣ��ž�װ���ڿ����Է�ֹ��ը����2����ɫ��ĩ����ɫ��ĩ����3��3CO+Fe2O3
| ||
��4��������̼��CO2+Ca��OH��2=CaCO3��+H2O����5����ֹδ��Ӧ��һ����̼��Ⱦ������
������������Ҫ����һ����̼����������Ӧʵ��װ�á�ʵ������һ����̼�Ļ�ѧ���ʺ���Ⱦ��֪ʶ���ѶȲ����������ݿα�����֪ʶ�����ش�

��ϰ��ϵ�д�
 ������ȫ��������ϵ�д�
������ȫ��������ϵ�д�
�����Ŀ
ij+2�۵Ľ�������������Ԫ�ص���������Ϊ40%����ý���Ԫ�ص����ԭ������Ϊ��������
| A��24 | B��40 | C��56 | D��65 |