��Ŀ����
��һ�ο����У��Ⱥ�������ͬѧ�ú���̼���Ƶ��Ȼ��ƹ������������ʵ�飮ʵ����ʦ�ṩ���Լ��У���������Ȼ�����Һ������������Һ����1��Ϊ�˵õ��������Ȼ�����Һ����һ��ͬѧȡһ�����������������ȫ���ܽ���ˮ��Ȼ�������ѡ�Լ������ɳ�����ǡ����ȫ��Ӧ����ˣ�������ҺΪ�Ȼ�����Һ�������ͬѧѡ����Լ���
��2��Ϊ�ⶨ�ù����������Ȼ��Ƶ������������ڶ���ͬѧȡ�ù�������26.5 g��ȫ���ܽ���ˮ�У�Ȼ�������һ����ѡ�Լ�����Ӧ����ҺpH����7��������������Ϊ2.2 g���ù���������̼���Ƶ���������Ϊ���٣�
��3��������ͬѧ���õ�һ��ͬѧ�����ᴿ�Ȼ��Ƶķ���������600 g��������Ϊ10%���Ȼ�����Һ����Ҫ��������������ٿˣ�
��������1�������Լ����г������ɣ�˵����������Ȼ�����Һ������������Һ�����ڼ�������������Һʱ�������������ƣ�����ʹ������Һ��ֻ���Ȼ��ƣ���˲���ѡ����������Һ��ֻ��ѡ�Ȼ�����Һ��
��2���������������ɣ���˿����жϼ�����Լ������ᣮ�������������������ȡ�������������������̼���������ᷴӦ�Ļ�ѧ����ʽ�����Լ�����ù���������̼���Ƶ�����������
��3���������Ƶ��Ȼ�����Һ���������������������Լ������������Һ�������Ȼ��Ƶ���������Щ�Ȼ�����������Դ��һ��������������ֱ���ṩ����һ���������������ڳ�ȥ����ʱ��Ӧ���ɣ�
��2���������������ɣ���˿����жϼ�����Լ������ᣮ�������������������ȡ�������������������̼���������ᷴӦ�Ļ�ѧ����ʽ�����Լ�����ù���������̼���Ƶ�����������
��3���������Ƶ��Ȼ�����Һ���������������������Լ������������Һ�������Ȼ��Ƶ���������Щ�Ȼ�����������Դ��һ��������������ֱ���ṩ����һ���������������ڳ�ȥ����ʱ��Ӧ���ɣ�
����⣺��1�����������֪�������Լ����������µ����ʣ����һ�Ҫ�г������ɣ�
������һҪ���ֻ�Т��Ȼ�����Һ��
��2����ԭ�������̼���Ƶ���������Ϊx��
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 44
26.5g?x 2.2g
=
��x=20%
��3��������600g10%�Ȼ�����Һ��Ҫԭ����������Ϊy��
��ȥ��Щ������е�̼����ʱ�����Ȼ��Ƶ�����Ϊz��
Na2CO3+CaCl2=CaCO3��+2NaCl
106 117
y?20% z
=
��z=
���������y?��1-20%��+
=600g��10%
���y��58.78g
�𣺣�1���ڣ�
��2��ԭ�������̼���Ƶ���������20%��
��3����Ҫԭ�����58.78g��
������һҪ���ֻ�Т��Ȼ�����Һ��
��2����ԭ�������̼���Ƶ���������Ϊx��
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 44
26.5g?x 2.2g
| 106 |
| 44 |
| 26.5g?x |
| 2.2g |
��3��������600g10%�Ȼ�����Һ��Ҫԭ����������Ϊy��
��ȥ��Щ������е�̼����ʱ�����Ȼ��Ƶ�����Ϊz��
Na2CO3+CaCl2=CaCO3��+2NaCl
106 117
y?20% z
| 106 |
| 117 |
| y?20% |
| z |
| 117?y?20% |
| 106 |
���������y?��1-20%��+
| 117?y?20% |
| 106 |
���y��58.78g
�𣺣�1���ڣ�
��2��ԭ�������̼���Ƶ���������20%��
��3����Ҫԭ�����58.78g��
������������Ҫ���麬�������ʵĻ�ѧ����ʽ������������������ļ��㣬�ѶȽϴ�
��������ԭ���������ʱ������Ĺؼ����ҳ�����ԭ�������������Һ���Ȼ��Ƶ�����������ϵ��
��������ԭ���������ʱ������Ĺؼ����ҳ�����ԭ�������������Һ���Ȼ��Ƶ�����������ϵ��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
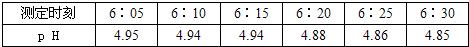
ijУ��ѧ����С����һ�λ�У�ȡ�ս����������ˮˮ����ÿ��5������pH�Ʋⶨһ��pH�����������±���
| �ⶨʱ�� | 6��05 | 6��10 | 6��15 | 6��20 | 6��25 | 6��30 |
| pH | 4.95 | 4.94 | 4.94 | 4.88 | 4.86 | 4.85 |
��1����ȡ����ˮ�Ƿ�Ϊ���ꣿ________����ǡ������ڲⶨ���ڼ䣬��ˮˮ������������ʱ��ı仯��________�����ǿ������������
��2��С�ָ���pH��ֽ�ⶨ��ˮ�����ȣ����Ƚ�PH��ֽ��ʪ�����ò�����պȡ������ˮ�ε�PH��ֽ�ϣ����������ɫ���Աȶ�������ʱ����õ�PHֵӦ��________���ƫ��ƫС�������䡱����
��3��Ϊ�˸��Ƴ��л������������д�ʩ�ܼ����������________������ţ���
���ƹ������Դ
�ڼ�����úֱ����ȼ��
����̭β������������
������ȼ���̻���������Ʋ��г�ʹ�����ϴ���
��ѧ����֮��ķ�Ӧ���������һ�λ�ѧ̽����У�ͬѧ�ǴӲ�ͬ�Ƕȶ�ϡ����������������Һ�ķ�Ӧ������̽����������벢��д���¿հף�
��1�������ʵ����֤ϡ����������������Һ�����˷�Ӧ
��һ��ͬѧ�����ʵ�鷽��һ��
�������������������Ϊʲôǿ��ֻ�в�õ�PH��7����֤����Ӧ�����ˣ�______
______��
��д��ϡ����������������Һ��Ӧ�Ļ�ѧ����ʽ______��
��2�����ʵ�飬̽��ϡ����������������Һ�ķ�Ӧ�Ƿ����
ʵ��������ҩƷ��10%�����ᡢ10%������������Һ���ձ����¶ȼơ�����������ͷ�ιܣ�
�������ʵ�鷽����ƣ�
��3��̽��ϡ����������������Һ�Ƿ�ǡ����ȫ��Ӧ
�ڶ���ͬѧ������ϡ����������������Һ��Ӧ��ʵ���Ϊ̽��ϡ����������������Һ�Ƿ�ǡ����ȫ��Ӧ��
�����ʵ�鷽������
��ʱ��ͬѧ�������������֤��ϡ����������������Һǡ����ȫ��Ӧ����ԭ����______��Ϊ�ˣ�����Ҫѡ��______����һ���Լ������ٽ���ʵ�鼴�ɣ�
����������
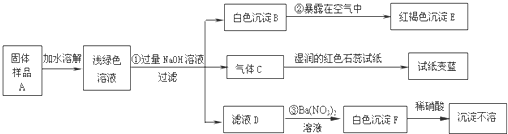
ij���½�һ��������һ�ֲ�Ʒ���ڡ����Ρ����ò�Ʒ��������������鵵Ȼ�ľ�ķ��ϣ�ij��ѧ������ȤС��̽���ò�Ʒ����ɣ����������µ�ʵ�飺����
�Իش��������⣺
��1��д���������ʵĻ�ѧʽ��C______��E______��F______
��2��д�����б仯�Ļ�ѧ����ʽ��
��______ ��______
��3������̽�����˵����ƷA�к��е�������______��
��1�������ʵ����֤ϡ����������������Һ�����˷�Ӧ
��һ��ͬѧ�����ʵ�鷽��һ��
| ʵ�鲽�� | ʵ������ | ���� |
| ����PH��ֽ�ⶨϡ�����PH�����μ�����������Һ����������ͬʱ�ⶨ���Һ��PH�� | PH��� ��� PH��7 | ϡ����������������Һ�ܷ�����Ӧ |
______��
��д��ϡ����������������Һ��Ӧ�Ļ�ѧ����ʽ______��
��2�����ʵ�飬̽��ϡ����������������Һ�ķ�Ӧ�Ƿ����
ʵ��������ҩƷ��10%�����ᡢ10%������������Һ���ձ����¶ȼơ�����������ͷ�ιܣ�
�������ʵ�鷽����ƣ�
| ʵ �� �� �� | ʵ������ | ���� |
| ϡ����������������Һ��Ӧ���� |
�ڶ���ͬѧ������ϡ����������������Һ��Ӧ��ʵ���Ϊ̽��ϡ����������������Һ�Ƿ�ǡ����ȫ��Ӧ��
�����ʵ�鷽������
| ʵ�鲽�� | ʵ������ | ���� |
| ȡ����������Ӧ�����Һ���Թ��У��μ�����ͭ��Һ�� | ��______ | ����������Һ���� |
| ��û���������� | ϡ����������������Һǡ����ȫ��Ӧ |
����������
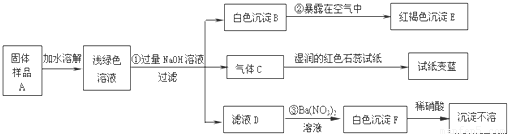
ij���½�һ��������һ�ֲ�Ʒ���ڡ����Ρ����ò�Ʒ��������������鵵Ȼ�ľ�ķ��ϣ�ij��ѧ������ȤС��̽���ò�Ʒ����ɣ����������µ�ʵ�飺����

�Իش��������⣺
��1��д���������ʵĻ�ѧʽ��C______��E______��F______
��2��д�����б仯�Ļ�ѧ����ʽ��
��______ ��______
��3������̽�����˵����ƷA�к��е�������______��