��Ŀ����
����Ŀ��ʵ���ҳ���������װ����������Ʊ�������ʵ�顣��ش���������
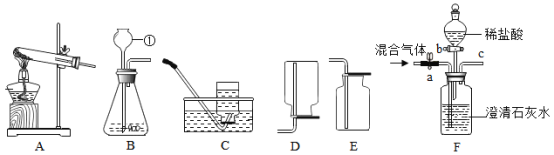
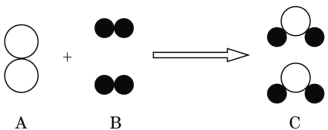
��1��ָ���������������:��_____��
��2������Aװ����һ�����ԵĴ������_____�����ø������װ�ü�����ɺ����ʵ�顣
��3����ʹ�ø��������ȡ����,Ӧѡ��Aװ��,�÷�Ӧ�Ļ�ѧ����ʽΪ_____��Ϊ��ֹ������ط�ĩ���뵼��,Ӧ��ȡ�Ĵ�ʩ��_____��
��4����CO2��CO��һ����̼���Ļ������ͨ��Fװ�ã�Һ��ҩƷ��������,ʵ�鿪ʼʱ�رջ���b��������a����ʱ�ӵ���c�ݳ���������Ҫ��һ����̼��һ��ʱ����ٹرջ���a������b������Һ©���е�ϡ���������ƿ�У���ʱ�ݳ���������Ҫ��_____���÷�Ӧ�Ļ�ѧ����ʽΪ_____�����ռ������壬Ӧѡ����ռ�װ����_____������ĸ����
���𰸡�����©�� Aװ���еĵ��������Թ�̫�� ![]() ���Թܿڷ�һ���� ������̼
���Թܿڷ�һ���� ������̼ ![]() E
E
��������
������ؼ�����������ء��������̺�������ϡ�����̼��������Ȼ��ơ�ˮ�Ͷ�����̼��
��1������������:�ٳ���©����
��2��Aװ����һ�����ԵĴ������Aװ���еĵ��������Թ�̫��������Ϊ������Ӧ¶����Ƥ���������ɡ����ø������װ�ü�����ɺ����ʵ�顣
��3��������ؼ�����������ء��������̺���������ʹ�ø��������ȡ������Ӧѡ��Aװ�ã��÷�Ӧ�Ļ�ѧ����ʽΪ![]() ��Ϊ��ֹ������ط�ĩ���뵼�ܣ�Ӧ��ȡ�Ĵ�ʩ�����Թܿڷ�һ������
��Ϊ��ֹ������ط�ĩ���뵼�ܣ�Ӧ��ȡ�Ĵ�ʩ�����Թܿڷ�һ������
��4����CO2��CO�Ļ������ͨ��Fװ�ã�Һ��ҩƷ����������ʵ�鿪ʼʱ�رջ���b������a��ƿ�й۲쵽�������dz����ʯ��ˮ����ǣ�������̼�ͳ���ʯ��ˮ��Ӧ����̼��ƺ�ˮ����ʱ�ӵ���c�ݳ���������Ҫ��һ����̼��һ����̼��ʯ��ˮ����Ӧ��һ��ʱ����ٹرջ���a������b������Һ©���е�ϡ�������ƿ�У�ϡ����ͷ�Ӧ���ɵ�̼��Ʒ�Ӧ�����ɶ�����̼����ʱ�ݳ���������Ҫ�Ƕ�����̼������������ְѶ�����̼�ͷų�������Ӧ��ϡ�����̼��������Ȼ��ơ�ˮ�Ͷ�����̼����Ӧ�Ļ�ѧ����ʽΪ
![]() ��������̼�ܶȱȿ�����������ˮ�����������ſ������ռ�����Ӧѡ����ռ�װ����E��
��������̼�ܶȱȿ�����������ˮ�����������ſ������ռ�����Ӧѡ����ռ�װ����E��

����Ŀ��С����ͬѧ��һ������գ���ҿ�����������⣬������ȼ�ղ�������Ȥ��
��������⣩С����������������о������һ�ư��̣����ư��̵ijɷ���ʲô��
����������裩����һ������������ȼ�ղ����Ķ�����̼��
�����������������ȼ�ղ�����__________��
��������������ʯ����������ɵ�ʯ�����������
���ռ�֤�ݣ���������
��1�������ɹ�������γɵģ�������СҺ���γɵģ�
��2��ʯ�����۵�ͷе㶼�ܵͣ�������Һ����������
��3������ȼ�ղ���ˮ�������¶ȸ���100��������̬��
��4��������̼����ɫ����ʹ����ʯ��ˮ����ǵ����塣
��ʵ����̽�����ڻ�ѧ��ʦ�İ����£�����չ����ʵ��̽����
�������� | ʵ������ | ���� |
ʵ��һ����������������һ���ڱ�Ϳ�г���ʯ��ˮ���ձ���ס���� | ����ʯ��ˮ����� | �����Ƕ�����̼�� ����һ���� |
ʵ������������������øɶ�����ձ����ڰ����� | �ձ���û�г���_____�����dz�����һЩ��ɫ�������� | ���̲���ˮ���� ����������� |
ʵ��������������������ȼ�ŵ�ľ���������� | �������±���ȼ | ������_______�� ���������� |
����������ͣ�
��1���������Ҫ�ɷ�������ʮ���飨![]() ���������������������ȼ��ʱ��ֻ���ɶ�����̼��ˮ����д���仯ѧ���ű���ʽ___________��
���������������������ȼ��ʱ��ֻ���ɶ�����̼��ˮ����д���仯ѧ���ű���ʽ___________��
��2��ʵ������֤�����̲����Ƕ�����̼����ʵ��һ����������ȷʵ�۲쵽ʯ��ˮ����ǣ�ԭ�������__________��
����˼�����ɣ��õ���ȷ�����Ժ�С����ͬѧ�Ƿ�˼��ʵ��̽�����̣����ֲ��þ���ʵ��̽��Ҳ�����ų�����һ�Ͳ������������________��
����Ŀ��ij�о���ѧϰС���ȡ��NaCl��Na2CO3����25 g���������Ƴ���Һ������������μ���������7.3%��ϡ���ᣬʹ������ȫ�ų������ռ��� 8.8 g CO2���塣
��1������ԭ������Na2CO3����������____________������ϡ�����������____________(д���������)��
��2���±�Ϊ�о���ѧϰС�����������ƵĻ��Һ�з�������μ���(�ӱ���)ϡ���������¼�IJ������ݡ�����������֪Na2CO3��HCl��Ӧ���Է��������У�Na2CO3��HCl=NaCl��NaHCO3��NaHCO3��HCl=NaCl��H2O��CO2����
������ɱ�����δ��IJ��֡�
ʵ����� | ÿ�β�����CO2������/g |
��һ������εμ�ϡ����100 g | ________ |
�ڶ�������εμ�ϡ����100 g | 8.8 |
����������εμ�ϡ����100 g | 0 |
�ڸ��ݱ�������������ϵ�л���CO2������(������)��������ϡ���������(������)�Ĺ�ϵͼ________��