��Ŀ����
ȡֻ���������Ȼ������ʵķ�ĩ״������Ʒ11.4g���ձ��У���μ���ϡ���������ٲ�������Ϊֹ��������ϡ���������Ϊ50g����Ӧ��Ƶ��ձ�����Һ������Ϊ57g���ٶ���Ӧ�����Ķ�����̼ȫ���ݳ���ˮ�����ӷ����Բ��ƣ���������ش��������⣺
��1�������漰���Ļ�ѧ��Ӧ����ʽΪ______��
��2�����������غ㶨�ɣ�����������зų�������̼������Ϊ______g����3�����㷴Ӧ����Һ�����ʵ�����������д����ϸ�ļ�����̣����ݾ�ȷ��l%����______��
�⣺
��1�����Ȼ��ƺ�̼���ƵĻ�����м���ϡ���ᣬ�Ȼ��ƺ������Ӧ��̼���ƺ����ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼���ʴ�Ϊ��Na2CO3+2HCl=2NaCl+H2O+CO2��
��2��11.4g+50g-57g=4.4g �ʴ�Ϊ��4.4
��3���⣺��μӷ�Ӧ��Na2CO3������Ϊx����Ӧ������NaCl������Ϊy
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 117 44
x y 4.4g

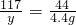
x=10.6g y=11.7g
�ʷ�Ӧ����Һ�����ʵ���������Ϊ��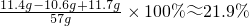
�𣺷�Ӧ����Һ�����ʵ���������Ϊ21.9%��
��������1�����ݸ��ֽⷴӦ�������������з�����
��2�����������غ㶨�ɣ���Ӧǰ���ձ������ʼ��ٵ�����Ϊ������̼��������
��3����Ӧ����Һ�����ʵ���������=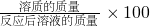 %����Һ��������֪������Ϊ�Ȼ��ƣ�����ԭ������е��Ȼ��ƣ����ݶ�����̼���������̼���Ƶ���������ԭ����������-̼���Ƶ�������Ϊԭ��������Ȼ��Ƶ��������ͷ�Ӧ���ɵ��Ȼ��ƣ����ݶ�����̼�������������
%����Һ��������֪������Ϊ�Ȼ��ƣ�����ԭ������е��Ȼ��ƣ����ݶ�����̼���������̼���Ƶ���������ԭ����������-̼���Ƶ�������Ϊԭ��������Ȼ��Ƶ��������ͷ�Ӧ���ɵ��Ȼ��ƣ����ݶ�����̼�������������
������������Ҫ������ѧ������йغ��������ʵĻ�ѧ��Ӧ�ļ��������������ؼ�����Ӧ����Һ�е����ʵ�����ʱ����Ҫ�������ɵ����ʣ���Ҫ����ԭ�������е����ʣ�
��1�����Ȼ��ƺ�̼���ƵĻ�����м���ϡ���ᣬ�Ȼ��ƺ������Ӧ��̼���ƺ����ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼���ʴ�Ϊ��Na2CO3+2HCl=2NaCl+H2O+CO2��
��2��11.4g+50g-57g=4.4g �ʴ�Ϊ��4.4
��3���⣺��μӷ�Ӧ��Na2CO3������Ϊx����Ӧ������NaCl������Ϊy
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 117 44
x y 4.4g

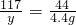
x=10.6g y=11.7g
�ʷ�Ӧ����Һ�����ʵ���������Ϊ��
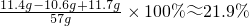
�𣺷�Ӧ����Һ�����ʵ���������Ϊ21.9%��
��������1�����ݸ��ֽⷴӦ�������������з�����
��2�����������غ㶨�ɣ���Ӧǰ���ձ������ʼ��ٵ�����Ϊ������̼��������
��3����Ӧ����Һ�����ʵ���������=
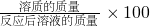 %����Һ��������֪������Ϊ�Ȼ��ƣ�����ԭ������е��Ȼ��ƣ����ݶ�����̼���������̼���Ƶ���������ԭ����������-̼���Ƶ�������Ϊԭ��������Ȼ��Ƶ��������ͷ�Ӧ���ɵ��Ȼ��ƣ����ݶ�����̼�������������
%����Һ��������֪������Ϊ�Ȼ��ƣ�����ԭ������е��Ȼ��ƣ����ݶ�����̼���������̼���Ƶ���������ԭ����������-̼���Ƶ�������Ϊԭ��������Ȼ��Ƶ��������ͷ�Ӧ���ɵ��Ȼ��ƣ����ݶ�����̼�������������������������Ҫ������ѧ������йغ��������ʵĻ�ѧ��Ӧ�ļ��������������ؼ�����Ӧ����Һ�е����ʵ�����ʱ����Ҫ�������ɵ����ʣ���Ҫ����ԭ�������е����ʣ�

��ϰ��ϵ�д�
�����Ŀ