��Ŀ����
20��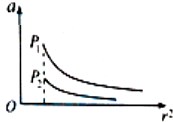 P1��P2Ϊ���ңԶ���������ǣ��뾶��ͬ��ͼ���������ʾ���Ƕ���Χ�ռ������������������ļ��ٶ�a���������ʾ���嵽�������ĵľ���r��ƽ�����������߷ֱ��ʾP1��P2��Χ�����a��r2�ķ��ȹ�ϵ����������˵�ĺ�������ͬ��������
P1��P2Ϊ���ңԶ���������ǣ��뾶��ͬ��ͼ���������ʾ���Ƕ���Χ�ռ������������������ļ��ٶ�a���������ʾ���嵽�������ĵľ���r��ƽ�����������߷ֱ��ʾP1��P2��Χ�����a��r2�ķ��ȹ�ϵ����������˵�ĺ�������ͬ��������| A�� | ����P1������������ٶȱ�P2��С | B�� | ����P1�ġ���һ�����ٶȡ���P2��С | ||
| C�� | ����P1����ת���ڱ�P2�Ĵ� | D�� | ����P1���ܶȱ�P2��Ҫ�� |
���� ����ţ�ٵڶ����ɵó����Ƕ���Χ�ռ������������������ļ��ٶ�a�ı���ʽ�����a��r2�ķ��ȹ�ϵ����ͼ��ó�P1��P2���������������������ṩ���������������������������Ƚϵ�һ�����ٶ��Լ����DZ�����������ٶȴ�С��ϵ��
��� �⣺����ţ�ٵڶ����ɣ����Ƕ���Χ�ռ������������������ļ��ٶ�Ϊ��a=$\frac{GM}{{r}^{2}}$��P1��P2�İ뾶��ȣ����a��r2�ķ��ȹ�ϵ����ͼ��ó�P1����������P2������������$��=\frac{M}{\frac{4��{R}^{3}}{3}}$������P1��ƽ���ܶȱ�P2�Ĵ�D��ȷ��
����$G\frac{Mm}{{R}^{2}}=m\frac{{v}^{2}}{R}$�ã���һ�����ٶ�v=$\sqrt{\frac{GM}{R}}$��P1����������P2��������������P1�ġ���һ�����ٶȡ���P2�Ĵ�B����
����$G\frac{Mm}{{R}^{2}}=mg$�ã����������������ٶ�g=$\frac{GM}{{R}^{2}}$��P1����������P2����������������P1������������ٶȱ�P2�Ĵ�A����
������Ŀ�������Ƚ������ǵ���ת���ڴ�С����C����
��ѡ��D��
���� ����Ҫ��Ҫ��һ���Ķ�ͼ��������ѧ�������������ͼ�ж���һЩ��Ϣ��ͨ��ͼ�ߣ���������������ɹ�ʽ��ţ�ٵڶ����ɵó�����������С��ϵ�ǽ������Ĺؼ���

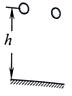 ��ͼ��ʾ��������ͬ������С���ͬһ�߶��ɾ�ֹͬʱ�ͷţ����ƿ�������������˵����ȷ���ǣ�������
��ͼ��ʾ��������ͬ������С���ͬһ�߶��ɾ�ֹͬʱ�ͷţ����ƿ�������������˵����ȷ���ǣ�������| A�� | �������С����ٶȴ� | B�� | �������С������� | ||
| C�� | ��������������ٶȱ仯һ���� | D�� | �������С�����ǰ˲����ٶȴ� |
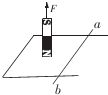 ��ͼ���⻬ƽ�н�������̶���ˮƽ���ϣ�����ɵ��������������ab��ֱ�����ڵ����Ϲ��ɻ�·��������F�����£���·�Ϸ������δ������¶���N������ֱ�����������˶����������˶�ij��ʱ��������F����WF���ų����Ե��������W1�������˷��ų�������W2�������Դ�������WG����·�в����Ľ�����ΪQ���������õĶ���ΪEk��������ѡ������ȷ���ǣ�������
��ͼ���⻬ƽ�н�������̶���ˮƽ���ϣ�����ɵ��������������ab��ֱ�����ڵ����Ϲ��ɻ�·��������F�����£���·�Ϸ������δ������¶���N������ֱ�����������˶����������˶�ij��ʱ��������F����WF���ų����Ե��������W1�������˷��ų�������W2�������Դ�������WG����·�в����Ľ�����ΪQ���������õĶ���ΪEk��������ѡ������ȷ���ǣ�������| A�� | ������еĵ�������Ϊa��b | B�� | Wl=W2 | ||
| C�� | W2-W1=Q | D�� | WF+WG=Ek+Q |
| A�� | �����ܵ��㶨��������ʱ�������˶�״̬�������ı� | |
| B�� | �����ܵ��ĺ���Ϊ��ʱ����һ�����ھ�ֹ״̬ | |
| C�� | �����ܵ���Ϊ��ĺ�������ʱ�������˶�״̬�ͷ����ı� | |
| D�� | ������˶�����һ���������ܵĺ����ķ�����ͬ |