��Ŀ����
��ӦCH3OH ( l )+ NH3( g ) = CH3NH2( g ) + H2O ( g )��ij�¶��Է����ҽ��У�����Ӧ����H��= 17kJ/mol������H��T��S��= 17kJ/mol�����ڸ÷�Ӧ���й�ϵ��ȷ���ǣ� ��
A. ��H ��0����H�CT��S��0 B. ��H ��0����H�CT��S��0
C. ��H ��0����H�CT��S��0 D. ��H ��0����H�CT��S��0
A
����:
��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
��֪ij�¶������ܱ������м���CH3OH���������¿��淴Ӧ��
2CH3OH��g��==CH3OCH3��g��+H2O��g������Ӧ��ijʱ�̲�ø���ֵ����ʵ���Ũ�ȣ�mol��L-1�����£������й�˵������ȷ���ǣ� ��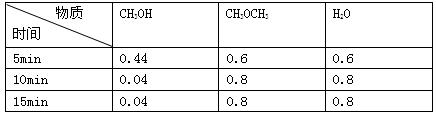
| A��10minʱv��=v�� |
| B��5minʱ�÷�Ӧ����v(CH3OH)=0.088mol��L-1��min-1 |
| C�����¶��´˷�Ӧ��ƽ�ⳣ��k=400 |
| D��ƽ��������������䣬���£�c(CH3OH)=0.06mol/L���÷�Ӧ��H<0 |