��Ŀ����
Ϊ�о���ù�أ�GA���������ᣨABA����ֲ�������������μ�ɺ����澳��������������ĵ������ã��о�����Ұ�������Ͻ���ת�����E��ʹ��ϸ����GA�������ͣ�ͻ���壩�������ͼ��

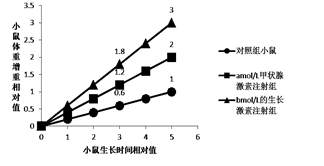
��1����ù�أ�GA���������ᣨABA����Ϊֲ�D�ء���������ֲ�����ڲ������Ӳ�����λ���䵽���ò�λ��������������_____________���� ����л�����������
��2����ͼ1��֪��һ��Ũ�ȵ�����Һ�����ɺ�������__________���Ͻ�Ĵ���ʸ��ߣ��ɴ˿�֪��GA����____����ߡ��͡��������������Ͻ����澳������������
��3����ͼ2��֪��Ұ�������Ͻ��ABA��������__________������ڡ����ڡ���ͻ�������Ͻ棬��ԭ������Ұ������ȣ�ͻ�������Ͻ���GA��ABA�ĺ����ֱ���__________________��
��4���ۺ�����������Ʋ⣬GA���������Ͻ�ֿ��澳���������йأ�GA����ͨ��Ӱ��ϸ���е�ABA�ź�;������ֲ������״̬��������ֲ���������������Ӧ�����仯�Ĺ����У�����ֲ�D�ز����ǹ��������ã����Ƕ��ּ���_____________________��
��1������Ӱ�죨�������ã� �л���
��2��ͻ���� ��
��3������ GA�����ͣ�ABA������
��4������ù�ͬ����
�������������
��1��ֲ�D������ֲ�����ڲ��������Ӳ�����λ���䵽���ò�λ����ֲ�������������������Ӱ������л��
��2����ͼ1��֪�����δ����ɺ�������ͻ�������Ͻ�Ĵ���ʸ��ߣ�ͻ�������ڵ����˻���E����GA�����ϵͣ��ɴ˿�֪��GA���������������Ͻ����澳������������
��3����ͼ2��֪������ABAŨ�ȵ����ӣ�ͻ����������ȷ���Ѹ�ٽ��ͣ�˵��Ұ�������Ͻ��ABA�������Ե���ͻ�������Ͻ棬��ԭ������Ұ������ȣ�ͻ�������Ͻ���GA�����ͣ�ABA�����ߡ�
��4���ۺ�����������Ʋ⣬GA���������Ͻ�ֿ��澳���������йأ�GA����ͨ��Ӱ��ϸ���е�ABA�ź�;������ֲ������״̬��������ֲ���������������Ӧ�����仯�Ĺ����У�����ֲ�D�ز����ǹ��������ã����Ƕ��ּ�������ù�ͬ���ڡ�
���㣺���⿼��ֲ�D�ص��ڵ��й�֪ʶ�����ڿ��鿼��ʶͼ�������������֡�ͼ���Լ���ѧ��ʽ�ȶ��ֱ�����ʽȷ����������ѧ��������ݣ���������ѧ֪ʶ��۵㣬ͨ���Ƚϡ��������ۺϵȷ�����ijЩ����ѧ������н��͡������������������жϻ�ó���ȷ�Ľ��ۡ�

ij��������ë����ɫ����ɫ�ز������������Ͱ�ɫ�������ͣ���A-a��B-b���Ե�λ������ơ�������������Ե�λ�������ڳ�Ⱦɫ���ϻ�������Ⱦɫ���ϣ��Լ����Ե�λ�����Ƿ���ϻ����������϶��ɡ��±�Ϊ��ػ���Ŀ����������ݱ��ش�
| | A���� | a���� | B���� | b���� |
| ������� | ����ɫ�� | ������ɫ�� | ��ֹɫ�صij��� | ��ʹɫ�س��� |
��2��������Ե�λ����λ�����Գ�Ⱦɫ���ϣ�����ëΪ��ɫ�ĸ���Ļ������� �֡�������ΪAaBb�Ĵơ����Ը����ӽ����Ӵ��ı������� ������֮���������Ϊ ��
��3��������Ե�λ����λ��ͬһ�Գ�Ⱦɫ���ϣ��������ΪAaBb�ĸ����������ϵ���ë��ɫ�����Ը����ӽ����Ӵ�����ë��ɫ����ë��ɫ=1:1�������ǽ��滥�������أ��ܷ�ȷ���û�����ΪAaBb�����һ��ϸ����A��a��B��b�ĸ������ڸö�ͬԴȾɫ���ϵľ���ֲ������ ����ܡ����ܡ�����
��4�����A��aλ�ڳ�Ⱦɫ���ϣ�B��bλ��ZȾɫ���ϣ���д���Ի�����ΪAaZBW�ĸ�����в⽻���Ŵ�ͼ�⡣
�̲ݻ�Ҷ������TMV�����̲�ҶƬ������Ե�Ӱ������ʵ����������ͼ���������ݶ��������������²�õġ���ش��������⣺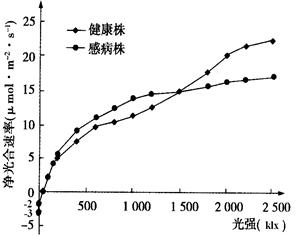
| ֲ������ | Ҷ����a������mg/g�� | Ҷ����b������mg/g�� |
| ������ | 2.108 | 0.818 |
| ��� | 1.543 | 0.604 |
��2�����ϱ����ݿ�֪��TMV��Ⱦ�̲ݺ����Ų����Ĵ�����ֳ��ϸ���е� ��ϸ�������ܵ��ƻ��� �ĺϳɼ��٣�ҶƬ�����ֻ��Ρ����̵�����
��3������ͼ��ʵ�����ݿ�֪������ǿС��1500 klxʱ���в���� ���ڽ����꣬˵����ʱ
����Ӱ�쾻������ʵ���Ҫ���ء� ����ǿ����1500 klxʱ���в����ʵ�ʹ������Ϊ ��mol�qm-2�qs-1��TMV�� �����¶Ըв��������õ����Ƹ�������
��4����TMV��RNA�복ǰ�ݲ����ĵ��������������һ��������IJ�����Ⱦ��Ҷ����Ҷ���ֵ��� ������Ⱦ��֢״����˵��TMV���Ŵ������� ��
�����ɭ���е���������֮һ����Ҫ��ֲ��Ŀ�֦��ҶΪʳ��Ϊ̽������ɭ�ֵ���������ã��о�����������ʵ�顣
��1��ɭ����̬ϵͳ�еĸ��������Ϊ____________������̬ϵͳ�ijɷֽǶȿ����������__________������̬ϵͳ�Ĺ��ܽǶȿ���������Ϊ�ٽ���___________________��
��2���о���ѡ��4�����ֵ�ҶƬ���˲�ͬ��������6��9�½���������ʵ�顣ÿ��ҶƬ�����������У���������Ͼ��ȣ�����Ŀ��ȵ������������������ͳ������ʳ����������������±���
��λ���������ƽ��ʳ����������mg/(g�qd)��
| ��ͬ���� | �ɹ��� | �� | ���� | ���� |
| δ�ֽ�Ҷ | 2.44 | 5.94 | 4.79 | 7.30 |
| ��ֽ�Ҷ | 7.20 | 9.42 | 8.71 | 5.23 |
�ھݱ�����������ֽ�Ҷ������������_________δ�ֽ�Ҷ�����������ڲ�ͬҶƬ�У���________________��Ϊϲ�á��ɴ�˵��____________________��Ӱ�������ʳƫ�õ���Ҫ���ء�
��3����������ʵ����������ں����ֺ��ɹ���������ֲһЩ�����������������������������࣬����������̬ϵͳ��______________��
 ����ϸ������������ش��������⣺
����ϸ������������ش��������⣺