��Ŀ����
�ش����Т�С��
���۹�Ӭ��XȾɫ�������ױ��е� Ӭ�������䴫����һ���е� Ӭ���۹�Ӭ�İ��ۻ���λ�� Ⱦɫ���ϣ� Ⱦɫ����û�иû���ĵ�λ���������������״���ְ����Ŵ���
����֪��Ӭ��ë�ͽ�ë��������״��X��YȾɫ����һ�Ե�λ������ƣ���ë����B���Խ�ë����ţ��b��Ϊ���ԣ����л����ͷֱ�ΪXBXB��XBYB��XbXb��XbYb�����ֹ�Ӭ��
��1��������Ҫ���������ֹ�Ӭ��ѡ���ױ���ͨ�������ӽ���ʹ���ջ�õĺ����Ӭ�У�����ȫ������Ϊ��ë������ȫ������Ϊ��ë�����һ���ӽ��ױ��У����ԵĻ������� �����ԵĻ������� ���ڶ����ӽ��ױ��У����ԵĻ������� �����ԵĻ������� �����ջ�õĺ���У���ë�۹�Ӭ�Ļ������� ����ë�ƹ�Ӭ�Ļ�������
��2��������Ҫ���������ֹ�Ӭ��ѡ���ױ���ͨ�������ӽ���ʹ���ջ�õĺ����Ӭ�д���ȫ������Ϊ��ë������ȫ������Ϊ��ë��Ӧ��ν���ʵ�飿�����ӽ�ʵ����Ŵ�ͼ���ʾ���ɣ�
���۹�Ӭ��XȾɫ�������ױ��е�
����֪��Ӭ��ë�ͽ�ë��������״��X��YȾɫ����һ�Ե�λ������ƣ���ë����B���Խ�ë����ţ��b��Ϊ���ԣ����л����ͷֱ�ΪXBXB��XBYB��XbXb��XbYb�����ֹ�Ӭ��
��1��������Ҫ���������ֹ�Ӭ��ѡ���ױ���ͨ�������ӽ���ʹ���ջ�õĺ����Ӭ�У�����ȫ������Ϊ��ë������ȫ������Ϊ��ë�����һ���ӽ��ױ��У����ԵĻ�������
��2��������Ҫ���������ֹ�Ӭ��ѡ���ױ���ͨ�������ӽ���ʹ���ջ�õĺ����Ӭ�д���ȫ������Ϊ��ë������ȫ������Ϊ��ë��Ӧ��ν���ʵ�飿�����ӽ�ʵ����Ŵ�ͼ���ʾ���ɣ�
���㣺�����Ŵ�
ר�⣺
��������Ӭ���Ա������ʽ��XY�ͣ��۹�Ӭ����Ⱦɫ�����ΪXY���ƹ�Ӭ����Ⱦɫ�����ΪXX����Ⱦɫ���ϵĻ�����Ƶ���״���Ա��������
���
�⣺���Ӭ���Ա������ʽ��XY�ͣ��۹�Ӭ����Ⱦɫ�����ΪXY������XȾɫ�������ױ��еĴ�Ӭ�������䴫����һ���еĴ�Ӭ���۹�Ӭ�İ��ۻ���λ��XȾɫ���ϣ�YȾɫ����û�иû���ĵ�λ���������������״���ְ����Ŵ���
��1���ӽ��������ǵ�F2����Ҫ��F2����ȫ������Ϊ��ë��XbYb��������ȫ������Ϊ��ë��XBX-����F1����ӦȫΪXbXb���ɴ��ṩ��������ֱ��ѡ���������б����л�������XBYb�ĸ��壬����ѡ��XBXB��XbYb�ӽ�������ԣ�XBYb���Ϳ����ˣ�
��2�����������ֹ�Ӭ��ѡ���ױ���ͨ�������ӽ���ʹ���ջ�õĺ����Ӭ�д���ȫ������Ϊ��ë������ȫ������Ϊ��ë��ʵ��������ƣ�

�ʴ��ǣ�
��� �� X Y
��1��XbYb XBXB XBYb XbXb XbYb XBXb
��2��

��1���ӽ��������ǵ�F2����Ҫ��F2����ȫ������Ϊ��ë��XbYb��������ȫ������Ϊ��ë��XBX-����F1����ӦȫΪXbXb���ɴ��ṩ��������ֱ��ѡ���������б����л�������XBYb�ĸ��壬����ѡ��XBXB��XbYb�ӽ�������ԣ�XBYb���Ϳ����ˣ�
��2�����������ֹ�Ӭ��ѡ���ױ���ͨ�������ӽ���ʹ���ջ�õĺ����Ӭ�д���ȫ������Ϊ��ë������ȫ������Ϊ��ë��ʵ��������ƣ�

�ʴ��ǣ�
��� �� X Y
��1��XbYb XBXB XBYb XbXb XbYb XBXb
��2��

���������⿼������Ŵ����֪ʶ����Ҫ������Ⱦɫ���ϵĻ�����Ƶ���״���Ա���أ���������ѧ�������������������Լ��Ŵ�ͼ�Ļ���������

��ϰ��ϵ�д�
 �����ܿ����ϵ�д�
�����ܿ����ϵ�д�
�����Ŀ
�����й��ݲ��������������κ����ⶨ�������У���ȷ���ǣ�������
| A���ݲ�������Ҫ������ˮ����������ˮ��������Ϊ4��l |
| B���ݲ�����ʱ�䳤�̻�Ӱ���������κ��������¶Ⱥ�ʳ�ε�������Ӱ���京�� |
| C���������κ����IJⶨ����Ϊ���ص���--�ữ--��ɫ--��ɫ |
| D������ɫ�����Ʒ����֪Ũ�ȱ�Һ����Ŀ��Ƚϣ��ɹ�����ݲ˵��������κ��� |
�����ܵ�ij�ֿ�ԭ�̼�����������ϸ���������ܵ�ͬ�ֿ�ԭ�ĵڶ��δ̼�������
| A������ϸ����ϸ�����ڳ���ʱ���̣����忹��Ũ������ |
| B������ϸ����ϸ�����ڳ���ʱ��䳤�����忹��Ũ������ |
| C������ϸ����ϸ�����ڳ���ʱ���̣����忹��Ũ�ȼ��� |
| D������ϸ����ϸ�����ڳ���ʱ�䲻�䣬���忹��Ũ�ȼ��� |

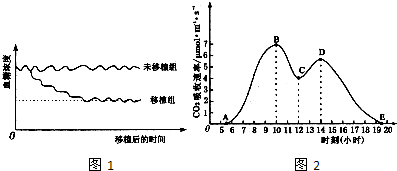
 ��С����̥��ϸ�������յ��ֻ���һ���ض���ϸ��������ΪMϸ�������ٽ�Mϸ����ֲ������ģ��С���ȵ�ϸ�����ض�ҩ���ƻ���С�����ڣ�Ȼ��С���Ѫ��Ũ�ȣ������ͼ��ʾ�����߱�ʾ����С���Ѫ��Ũ��ֵ������ش�������⣺
��С����̥��ϸ�������յ��ֻ���һ���ض���ϸ��������ΪMϸ�������ٽ�Mϸ����ֲ������ģ��С���ȵ�ϸ�����ض�ҩ���ƻ���С�����ڣ�Ȼ��С���Ѫ��Ũ�ȣ������ͼ��ʾ�����߱�ʾ����С���Ѫ��Ũ��ֵ������ش�������⣺