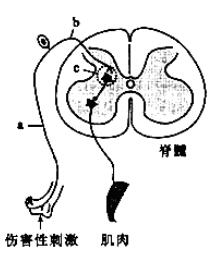
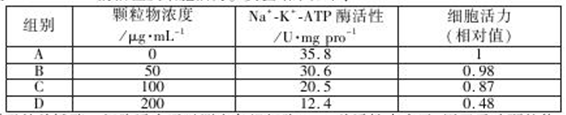
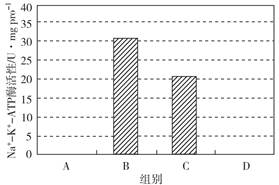
��Ŀ����
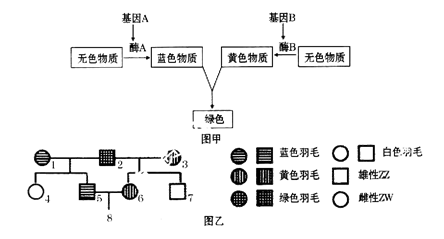
ɽ���Ա������ʽΪXY�͡������ϵ��ͼ��ʾ��ɽ��ij����״���Ŵ���ͼ����ɫ��ʾ������״�ı����ߡ�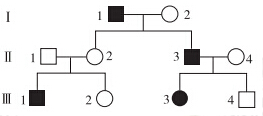
��֪����״��һ�Ե�λ������ƣ��ڲ�����Ⱦɫ�����ͻ���ͻ��������£��ش��������⣺
��1����ϵ��ͼ�Ʋ⣬����״Ϊ___________������ԡ������ԡ�����״��
��2��������Ƹ���״�Ļ����λ��YȾɫ���ϣ�����YȾɫ���ϻ�����Ŵ����ɣ��ڵ�III���У������Ͳ����ϸû����Ŵ����ɵĸ�����___________��������ţ���
��3�������Ƹ���״�Ļ����λ��XȾɫ���ϣ���ϵ��ͼ��һ�����Ӻ��ӵĸ�����___________��������ţ����������Ӻ��ӵĸ�����___________��������ţ���
��1�����ԣ�2����-1����-3�͢�-4��3����-2����-2����-4 ��-2
����

 �ܿ�����ĩ��̾�ϵ�д�
�ܿ�����ĩ��̾�ϵ�д�ֲ��ո��е���ά�ؿɱ�ijЩ����ֽ⣬�ش��������⣺
��1���ֽ�ո�����ά�ص������ܷ�����ά��ø����ø����3�������ɵĸ���ø��������������ø�ɽ� �ֽ�� ��
��2���ں���ά�ص��������м���չ��죨CR��ʱ��CR������ά���γ� ɫ������ú���CR�ĸ���������������ά�طֽ��ʱ���������ϻ�����Ըþ��ľ���Ϊ���ĵ� ��
��3��Ϊ�Ӹ�����ά�ص������з�������ά�طֽ���ĵ����䣬ijͬѧ����˼ס��������������ɷּ��±�����
| | ��ĸ�� | ���� | ���� | ��ά�ط� | ��֬ | CR��Һ | ˮ |
| �������� | + | + | + | + | - | + | + |
| �������� | + | + | + | - | + | + | + |
�ݱ��жϣ��������� ����ܡ����ܡ������ڷ���ͼ�����ά�طֽ����ԭ���� ���������� ����ܡ����ܡ������ڷ���ͼ�����ά�طֽ����ԭ���� ��
����A��B�����ܱա�������̬ƿ����������ɺ����������±���һ��ʱ�����Aƿ����̬ϵͳ��Bƿ�ȶ���
| ��̬ƿ��� | ���� | ���������ɳ | �������� | ˮ�� | ���ζ��� |
| A | + | + | + | + | + |
| B | - | + | + | + | + |
��ش�
��1��Aƿ������������ࡢˮ�ݺ��ζ��ﹲͬ�����һ�� �����������ˮ�ݵ��ּ��ϵΪ ��
��2��һ��ʱ���Bƿ�и���������Ⱥ�ܶ� ��ԭ���� �����ζ������Ⱥ�ܶ� ��ԭ���� ��
��3����̬ϵͳ�зֽ��ߵ������� ��