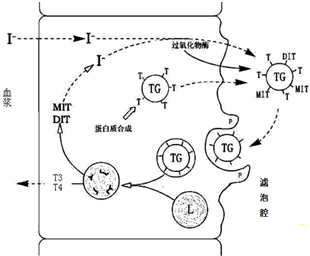
��Ŀ����
2���������ġ�����NH4+��PO43-��K+��Ca2+��ͬ����100mL����ˮ�У��ٷ���һЩ���ʵ�ˮ�����⣬һ��ʱ��ⶨ��Һ�������������Ӻ�ˮ�ĺ����仯�����±�����ʵ��������˵����������| ��Ŀ | H2O | NH4+ | K+ | Ca2+ | PO43- |
| ������ | 2% | 23% | 12% | 37% | 14% |
| A�� | ����ˮ�����պͶԿ������ӵ����������������Ĺ��� | |
| B�� | ���Կ������ӵ����ղ��ǵȱ������е� | |
| C�� | ��ϸ�����տ������ӵķ�ʽ���������� | |
| D�� | �����տ������ӵĹ��̲����¶ȵ�Ӱ�� |
���� ��������ʼʱ���������ӵ�Ũ����ͬ��һ��ʱ�����Һ��ˮ�ֺ������䣬���������ӵĺ����в�ͬ�̶ȵļ��٣�˵��ˮ������ˮ�ֵ����պͶԿ���Ԫ�ص�������������Զ����Ĺ��̣��Ҹ��Կ������ӵ����ղ��ǵȱ������еģ��ݴ˴��⣮
��� �⣺A������һ��ʱ�����Һ���������Ӻ�ˮ�ĺ����仯��֪��ˮ������ˮ�ֵ����պͶԿ���Ԫ�ص��������������̣����ǿ������ӱ����ܽ���ˮ����ܱ�ϸ�����գ��ʸ���ˮ�ֵ����պͶԿ���Ԫ�ص����ղ�����ȫ�����Ĺ��̣�A����
B��4������ԭ��Ũ����ͬ��һ���Ũ���в��죬˵�����Կ������ӵ����ղ��ǵȱ������еģ�B��ȷ��
C�������տ������ӵķ�ʽȷʵ���������䣬���ӱ��еò����ý��ۣ�C����
D�����ܴӱ��������еó������տ������ӵĹ����Ƿ����¶ȵ�Ӱ����һ���ۣ�D����
��ѡ��B��
���� ������ͼ��������ֲ���ˮ�ֵ����պ����ü�����Ӫ����Ҫ����ʶ��ֲ������ˮ�ַ�ʽ�����������̣�ʶ�ǿ���Ԫ�ص����ա���������ã��ܷ����������ݵó���ȷ���ۣ��ܽ����ѧ��֪ʶȷ�жϸ�ѡ����ڿ���ʶ�Ǻ������εĿ��飮

��ϰ��ϵ�д�
�����Ŀ
7��ij���Ӭ�ij����״����������λ������ƣ�������O��Բ��R����Բ��S���������������ױ����ӽ���ϣ����ǵĺ���������±�����ݱ��ش��������⣮
��1�����ӽ����2�����жϿ��ƹ�Ӭ�����״�Ļ���λ��XȾɫ���ϣ��һ���O��R��S֮�����������Թ�ϵΪ���á�������ʾ����S��O��R��
��2���ӽ����1��F1���۹�Ӭ��������õ���F2��F2����������F3���У������γ���Ӭ�������γ����Ӭ�ı���Ϊ7��6�����ӽ����1��F1��Ӭ�����ӽ����3��F1��Ӭ������䣬�����Ϻ��Ⱥ����S�Ļ���Ƶ��Ϊ���÷�����ʾ��$\frac{1}{3}$��
��3��Ϊȷ��ij��Բ�γ���Ӭ�Ļ����ͣ�ѡһֻ��Բ�γ����Ӭ��֮���䣮�������Ӭ��Լ��һ��������Ϊ�����γ����ô�Ӭ�Ļ�����ΪXSX�㣮
| �ӽ���� | �ױ� | F1 | ||
| ��Ӭ | ��Ӭ | ��Ӭ | ��Ӭ | |
| 1 | ������ | Բ�� | ������ | ������ |
| 2 | Բ�� | ������ | ������ | Բ�� |
| 3 | ������ | ��Բ�� | ��Բ�� | ������ |
��2���ӽ����1��F1���۹�Ӭ��������õ���F2��F2����������F3���У������γ���Ӭ�������γ����Ӭ�ı���Ϊ7��6�����ӽ����1��F1��Ӭ�����ӽ����3��F1��Ӭ������䣬�����Ϻ��Ⱥ����S�Ļ���Ƶ��Ϊ���÷�����ʾ��$\frac{1}{3}$��
��3��Ϊȷ��ij��Բ�γ���Ӭ�Ļ����ͣ�ѡһֻ��Բ�γ����Ӭ��֮���䣮�������Ӭ��Լ��һ��������Ϊ�����γ����ô�Ӭ�Ļ�����ΪXSX�㣮
10��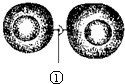 ��ͼ��ʾ��������ϸ����ϸ��Ĥ�Ӵ�����Ϣ��һ��ϸ�����ݸ���һ��ϸ���������йظ�ͼ��˵������ȷ���ǣ�������
��ͼ��ʾ��������ϸ����ϸ��Ĥ�Ӵ�����Ϣ��һ��ϸ�����ݸ���һ��ϸ���������йظ�ͼ��˵������ȷ���ǣ�������
 ��ͼ��ʾ��������ϸ����ϸ��Ĥ�Ӵ�����Ϣ��һ��ϸ�����ݸ���һ��ϸ���������йظ�ͼ��˵������ȷ���ǣ�������
��ͼ��ʾ��������ϸ����ϸ��Ĥ�Ӵ�����Ϣ��һ��ϸ�����ݸ���һ��ϸ���������йظ�ͼ��˵������ȷ���ǣ�������| A�� | �ٱ�ʾ��Ĥ��ϵ��źŷ��� | |
| B�� | ͼʾ�ɱ�ʾ���Ӻ���ϸ��֮���ʶ��ͽ�� | |
| C�� | ͼʾ�ɱ�ʾ�ȵ�ϸ�����ڵ��ȵ��������ڰ�ϸ�� | |
| D�� | ͼʾ���ɱ�ʾ�ߵ�ֲ��ϸ��֮�����ʽ�������Ϣ�����ķ�ʽ |

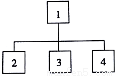
 �㶹�ӽ�����3�Ե�λ������Զ����Ŵ��������£����Ӵ������Ͳ�ͬ��2���ױ��ĸ�����ռȫ���Ӵ���
�㶹�ӽ�����3�Ե�λ������Զ����Ŵ��������£����Ӵ������Ͳ�ͬ��2���ױ��ĸ�����ռȫ���Ӵ���