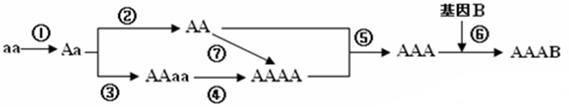
��Ŀ����
���������ܵ�������С·�Զ��Ǵ�Ƭ�ݵ�,�ж��ֶ�����Ϣ�� ��ͼ��ij������̬ϵͳʳ��������Ҫ���֡�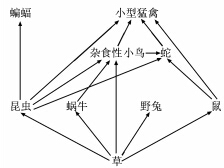
��ش���������:
(1)��ʳ������,С�����ݷֱ��� Ӫ������
(2)�����ڵ�С�����������ʮ�־���,���ַ�Ӧ���� ���䡣
(3)������Ա������̬ϵͳ�� Ϣ���ݵ��ص�,�ͷ�ѵ��������ӥ������С��
(4)Ϊ�˽�һ������,ij�����Ȳ���ԭ���Ӳ�,���������˳�������Եġ������ұ,������С���Ϊ���١��Ժ������ұ���Ӳݡ���ɱұ,��������Ⱥ��ı仯�������� ���档
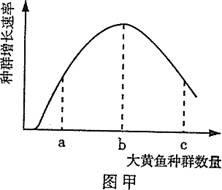
(5)Ϊ�˽��������ţ�ܶ�,����������Ա�������������������ͼ��ʾ(ͼ����Ӱ��Ϊˮ��С·,������Ϊ�ݵ�),������������� (�����)��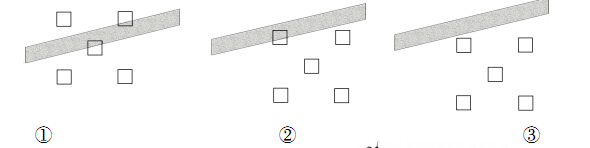
������ij����������ţ�ֲ�ʾ��ͼ����,����������ţ��Ӧ��Ϊ ����
��1�����������ġ����� ��2������ ��3����Ϊ ��4������ ��5���� 6 ��7
����

 �Ķ��쳵ϵ�д�
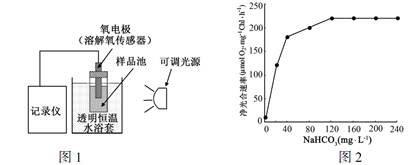
�Ķ��쳵ϵ�д�С����������ָʳ��������IJ��ˮ�����ε�ͨ��С����Ƥϸ������ѪҺ���ܰ͵Ĺ��̡�0.9%��NaCl��Һ�����õ���Һ��ѹ��ȵ�������ˮ��ijͬѧ����������״̬���õ�һ���ſ�С�������ɼס��ҡ��������ĸ�������ͬ��������ȵij�����ѪҺѭ����������������ʵ�飬ʵ�鲽���ʵ���������
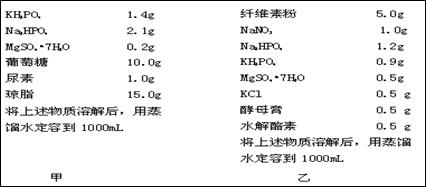
 ʵ����� ʵ�����ʵ�鲽�� | �� | �� | �� | �� |
| ����ע���������Һ�� ʹ���ӯ | 0.7��NaCl 10mL | 0.9��NaCl 10mL | 1.1��NaCl 10mL | 0.9��NaCl + ��Na+���� �������Ƽ���10mL |
| ά���������°�Сʱ�� �⳦����NaCl��Һ���� | 1mL | 3mL | 5mL | 9.9mL |
��1��ʵ�鿪ʼʱ��ˮ�ڳ�ǻ��ѪҺ֮����ƶ������ǣ������ ������� ������һ������ˮ���ӵ��˶������Ǵ���ҺŨ�� ��������ҺŨ�� ������ʵ����ˮ���ӵ������ƶ����̳�Ϊ ��
��2���Ƚ��ҺͶ���ʵ���������Ʋ�С������Na+ʱ����Ҫ �IJ��롣


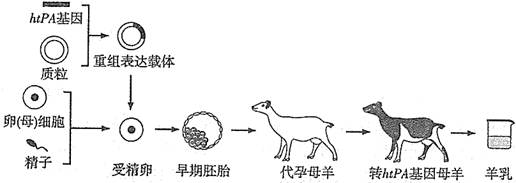
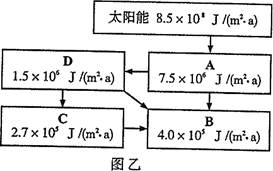
 ���������ṩ��Դ��������
���������ṩ��Դ��������