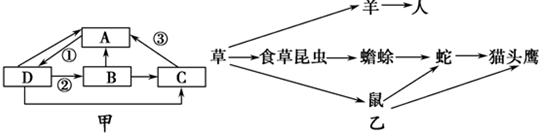
��Ŀ����
ij�ƣ�XX���ۣ�XY������ֲ���Ҷ������Ҷ��խҶ�������ͣ���һ�Ե�λ������ƣ����������ӽ�ʵ�飬������±����ݱ���������ȷ���ǣ�������
| �ӽ���� | �״������� | �Ӵ������ͼ����� | ||
| ���� | ĸ�� | ���� | ���� | |
| 1 | ��Ҷ | ��Ҷ | ��Ҷ234 | ��Ҷ119��խҶ122 |
| 2 | խҶ | ��Ҷ | ��Ҷ83��խҶ78 | ��Ҷ79��խҶ80 |
| 3 | ��Ҷ | խҶ | ��Ҷ131 | խҶ127 |
| A�����ݵ�1��ʵ�飬�����ж���ҶΪ���ԣ�խҶΪ���� | ||
| B�����ݵ�1����3��ʵ�����ȷ��Ҷ�λ���λ��XȾɫ���� | ||
C���õ�1����Ӵ���Ҷ������խҶ�����ӽ������խҶֲ��ռ
| ||
| D���õ�3����Ӵ���Ҷ��������Ҷ�����ӽ�����������ͱ���Ϊ1��2��1 |
���㣺�����Ŵ���ʵ���е�Ӧ��
ר�⣺
��������1��ʵ�飬���ݡ���������Ϊ���ԡ�������֪��ҶΪ���ԣ�խҶΪ���ԣ��Ӵ��������Ҷ����Ҷ��խҶ��Ŀ�����ȣ�������Ҷ��խҶһ����״���Ա��йأ���λ��XȾɫ���ϣ����÷��붨�ɼ������������Ӵ�խҶ����Ҷ�ĸ��ʣ�
���
�⣺
A����1��ʵ�飬���ݡ���������Ϊ���ԡ�������֪��ҶΪ���ԣ�խҶΪ���ԣ���A��ȷ��
B�����ݵ�1����3��ʵ���Ӵ��������Ҷ����Ҷ��խҶ��Ŀ�����ȣ�˵��Ҷ�����Ա��йأ�������������Ҷ����խҶ������ȷ��Ҷ�λ���λ��XȾɫ���ϣ���B��ȷ��
C������Ҷ�λ���ΪA��a����1��ʵ���У����Ӵ���������Ҷ����խҶ������֪ĸ��������ΪXAXa��������Ҷ������ΪXAY����һ����֪�Ӵ�����Ļ�����Ϊ1/2XAXA��1/2XAXa���Ӵ���Ҷ������խҶ�����ӽ������խҶֲ��ռ1/2��1/2=1/4����C��ȷ��
D������Ҷ�λ���ΪA��a����3��ĸ���������ΪXAY��ĸ���Ļ�����ΪXaXa�������Ӵ�������ΪXAXa��XaY���Ӵ���Ҷ��������Ҷ�����ӽ�����������ͱ���Ϊ�Ӵ��ӽ������XAXA��XAXa��XAY��XaY=1��1��1��1����D����
��ѡD��
A����1��ʵ�飬���ݡ���������Ϊ���ԡ�������֪��ҶΪ���ԣ�խҶΪ���ԣ���A��ȷ��
B�����ݵ�1����3��ʵ���Ӵ��������Ҷ����Ҷ��խҶ��Ŀ�����ȣ�˵��Ҷ�����Ա��йأ�������������Ҷ����խҶ������ȷ��Ҷ�λ���λ��XȾɫ���ϣ���B��ȷ��
C������Ҷ�λ���ΪA��a����1��ʵ���У����Ӵ���������Ҷ����խҶ������֪ĸ��������ΪXAXa��������Ҷ������ΪXAY����һ����֪�Ӵ�����Ļ�����Ϊ1/2XAXA��1/2XAXa���Ӵ���Ҷ������խҶ�����ӽ������խҶֲ��ռ1/2��1/2=1/4����C��ȷ��
D������Ҷ�λ���ΪA��a����3��ĸ���������ΪXAY��ĸ���Ļ�����ΪXaXa�������Ӵ�������ΪXAXa��XaY���Ӵ���Ҷ��������Ҷ�����ӽ�����������ͱ���Ϊ�Ӵ��ӽ������XAXA��XAXa��XAY��XaY=1��1��1��1����D����
��ѡD��
��������Ҫ������Է��붨�ɣ������ͺͱ����͵ȿ�������⣮���ڿ��鿼����������ѧ֪ʶ��Ҫ�㣬����֪ʶ���������ϵ��������

��ϰ��ϵ�д�
�����Ŀ
�����йط������ڵ������У�����ȷ���ǣ�������
| A��ʳ�����в�ʳ��ϵ��ά����Ҫ����Ⱥ���ڵķ������� |
| B����״�ټ��ء��Լ��ء��ȵ��ض�����ͨ����������ֱ�������������� |
| C��������������̬ϵͳ���ձ���ڣ�������̬ϵͳ���ҵ��������Ļ��� |
| D����ϸ���е�ATP��������ʱ��ATP�����ATP�ϳ�ø�����ã������ڸ����� |
�ڹ�����õİ���Ӧ��û�����ĵ������ǣ�������
| A����̼�������ø |
| B��H2O��CO2 |
| C��[H]��ATP |
| D����̼���������̼������ |
���˲ⶨ��A��B��C��D����ֲ�����ڶ���ø�Ļ������¶ȵĹ�ϵ���������ͼ��ʾ������ͼ����ʾ����Ϣ������Ϊ��25�������¾���������ǿ������Ͷ��¶���Ӧ��Χ��������ֱ��ǣ�������
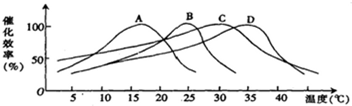

| A��B��D | B��B��C |
| C��B��A | D��A��C |
�����й�ϸ���ṹ������ȷ���ǣ�������
| A���������ǵ����ʵġ�װ����������ɵ����ʺ�mRNA��� |
| B����ø���ں��ж���ˮ��ø����ɱ������ϸ���IJ����� |
| C������������и��������ϸ����˿�ֹ����з�����Ҫ���� |
| D����ĸ��ϸ��������Ⱦɫ�壬���л�������������������� |
 С��߸ˣ�A�����ˣ�a��Ϊ���ԣ�������B�����в���b��Ϊ���ԣ���ͼ��ʾ�������˿���Ʒ�ֵļ���;�����������˵����ȷ���ǣ�������
С��߸ˣ�A�����ˣ�a��Ϊ���ԣ�������B�����в���b��Ϊ���ԣ���ͼ��ʾ�������˿���Ʒ�ֵļ���;�����������˵����ȷ���ǣ�������| A�����̢ٵ�ԭ���ǻ���ͻ�䣬����ŵ����������ڶ� | ||
| B�����̢�ʹ�õ��Լ�����ˮ���أ�����˿���Ѽ��ڷ������� | ||
| C�����̢�Ϊ���������֣������������������� | ||
D���ܹ��̵��Ӵ��д�������ռ������
|
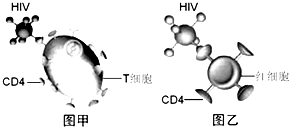 ����̬ѧͨѶ������������Ү³��ѧ������ѧ�ҳƣ��������ڴ��½��������յ��ܵ�������������Ϣ�ػ����������������о�����������������Ⱥ���յ���������ϸ���У��Է�ֹ����������ֳ���÷�����������սʤ�ܶ༲������AIDS������ϣ����
����̬ѧͨѶ������������Ү³��ѧ������ѧ�ҳƣ��������ڴ��½��������յ��ܵ�������������Ϣ�ػ����������������о�����������������Ⱥ���յ���������ϸ���У��Է�ֹ����������ֳ���÷�����������սʤ�ܶ༲������AIDS������ϣ����