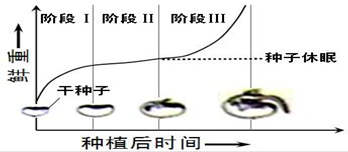
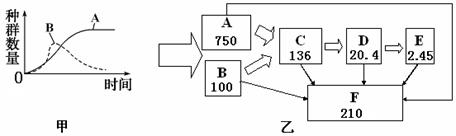
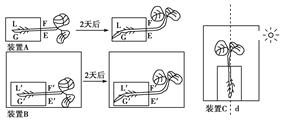
ΧβΡΩΡΎ»ί
ΫΒΗΤΥΊ «“Μ÷÷ΕύκΡάύΦΛΥΊΘ§ΝΌ¥≤…œ”Ο”Ύ÷ΈΝΤΙ«÷ ηΥ…÷ΔΒ»ΓΘ»ΥΒΡΫΒΗΤΥΊΜν–‘ΚήΒΆΘ§ΑκΥΞΤΎΫœΕΧΓΘΡ≥ΩΤ―ßΜζΙΙΈΣΝΥ―–ΖΔ“Μ÷÷Μν–‘ΗΏΓΔΑκΥΞΤΎ≥ΛΒΡ–¬–ΆΫΒΗΤΥΊΘ§¥”‘ΛΤΎ–¬–ΆΫΒΗΤΥΊΒΡΙΠΡή≥ωΖΔΘ§ΆΤ≤βœύ”ΠΒΡΆ――θΚΥή’Υα–ρΝ–Θ§≤Δ»ΥΙΛΚœ≥…ΝΥΝΫΧθ72ΗωΦνΜυΒΡDNAΒΞΝ¥Θ§ΝΫΧθΝ¥Ά®Ιΐ18ΗωΦνΜυΕ‘–Έ≥…≤ΩΖ÷ΥΪΝ¥DNAΤ§ΕΈΘ§‘Όάϊ”ΟKlenowΟΗ≤ΙΤΫΘ§ΜώΒΟΥΪΝ¥DNAΘ§Ιΐ≥Χ»γœ¬ΆΦΓΘ
‘Ύ¥ΥΙΐ≥Χ÷–ΖΔœ÷Θ§Κœ≥…Ϋœ≥ΛΒΡΚΥή’ΥαΒΞΝ¥“Ή≤ζ…ζ»± ßΦνΜυΒΡœ÷œσΓΘΖ÷ΈωΜΊ¥πœ¬Ν–Έ ΧβΘΚ
(1)KlenowΟΗ «“Μ÷÷________ΟΗΘ§Κœ≥…ΒΡΥΪΝ¥DNA”–________ΗωΦνΜυΕ‘ΓΘ
(2)ΜώΒΟΒΡΥΪΝ¥DNAΨ≠EcoRΔώ( Ε±π–ρΝ–ΚΆ«–ΗνΈΜΒψΓΣGΓΐAATTCΓΣ)ΚΆBamHΔώ( Ε±π–ρΝ–ΚΆ«–ΗνΈΜΒψΓΣGΓΐGATCCΓΣ)ΥΪΟΗ«–Κσ≤ε»κΒΫ¥σ≥ΠΗΥΨζ÷ ΝΘ÷–Θ§…Η―ΓΚ§÷ΊΉι÷ ΝΘΒΡ¥σ≥ΠΗΥΨζ≤ΔΫχ––DNA≤β–ρ―ι÷ΛΓΘ
ΔΌ¥σ≥ΠΗΥΨζ «άμœκΒΡ ήΧεœΗΑϊΘ§’β «“ρΈΣΥϋ_______________________________________________________________________________
_______________________________________________________ΓΘ
ΔΎ…ηΦΤEcoRΔώΚΆBamHΔώΥΪΟΗ«–ΒΡΡΩΒΡ «_______________________________________________________________________________
_____________________________________________________ΓΘ
Δέ“ΣΫχ––÷ΊΉι÷ ΝΘΒΡΦχΕ®ΚΆ―Γ‘ώΘ§–η“Σ¥σ≥ΠΗΥΨζ÷ ΝΘ÷–Κ§”–________ΓΘ
(3)Ψ≠DNA≤β–ρ±μΟςΘ§Ήν≥θΜώΒΟΒΡΕύΗω÷ΊΉι÷ ΝΘΘ§ΨυΈ¥ΖΔœ÷Άξ»Ϊ’ΐ»ΖΒΡΜυ“ρ–ρΝ–Θ§ΉνΩ…ΡήΒΡ‘≠“ρ «
______________________________________________________
______________________________________________________ΓΘ
(4)…œ ω÷Τ±ΗΗΟ–¬–ΆΫΒΗΤΥΊΘ§‘Υ”ΟΒΡœ÷¥ζ…ζΈοΙΛ≥ΧΦΦ θ «________ΓΘ
(1)DNAΨέΚœΟΗΓΓ126
(2)ΔΌΖ±÷≥ΩλΓΔΒΞœΗΑϊΓΔ“≈¥ΪΈο÷ œύΕ‘Ϋœ…ΌΓΓ
ΔΎ±Θ÷ΛΡΩΒΡΜυ“ρΚΆ‘ΊΧεΕ®œρΝ§Ϋ”(ΜρΖά÷ΙΡΩΒΡΜυ“ρΚΆ‘ΊΧε‘ΎΟΗ«–Κσ≤ζ…ζΒΡΡ©ΕΥΖΔ…ζ»Έ“βΝ§Ϋ”)Δέ±ξΦ«Μυ“ρ
(3)Κœ≥…ΒΡΚΥή’ΥαΒΞΝ¥»‘Ϋœ≥ΛΘ§≤ζ…ζ»± ßΦνΜυΒΡœ÷œσ
(4)ΒΑΑΉ÷ ΙΛ≥Χ
ΫβΈω

 ΧλΧλœρ…œ“Μ±ΨΚΟΨμœΒΝ–¥πΑΗ
ΧλΧλœρ…œ“Μ±ΨΚΟΨμœΒΝ–¥πΑΗ –Γ―ß…ζ10Ζ÷÷””Π”ΟΧβœΒΝ–¥πΑΗ
–Γ―ß…ζ10Ζ÷÷””Π”ΟΧβœΒΝ–¥πΑΗΘ®15Ζ÷Θ©÷≤ΈοΒΡ…ζ≥ΛΖΔ”ΐ «”…Εύ÷÷ΦΛΥΊœύΜΞ–≠ΒςΓΔΙ≤Ά§ΒςΫΎΒΡΓΘœ¬±μ «÷≤Έο≤ΜΆ§≤ΩΈΜΗς÷÷ΦΛΥΊΒΡœύΕ‘≈®Ε»Θ§«κΖ÷ΈωΜΊ¥πΘΚ
| ≤ΩΈΜ | ΦΛΥΊΒΡœύΕ‘≈®Ε» | |||
| …ζ≥ΛΥΊ | ≥ύΟΙΥΊ | œΗΑϊΖ÷Ν―ΥΊ | Ά―¬δΥα | |
| ΨΞΦβ | +++ | +++ | +++ | - |
| ”Ή“Ε | +++ | +++ | - | - |
| …λ≥ΛΨΞ | ++ | ++ | - | - |
| ≤ύ―Ω | + | + | - | - |
| ≥… λ“Ε | + | + | - | +++ |
| +++±μ ΨΚ§ΝΩΗΏΘΜ++±μ ΨΚ§ΝΩ÷–Β»ΘΜ+±μ ΨΚ§ΝΩΒΆΘΜ-±μ ΨΈό | ||||
Θ®1Θ©ΡΩ«ΑΙΪ»œΒΡ÷≤ΈοΦΛΥΊΙ≤”–Έε¥σάύΘ§≥ΐ…œ±μ÷–ΒΡΥΡ¥σάύΆβΘ§÷≤ΈοΧεΡΎΜΙ”–“Μάύ÷≤ΈοΦΛΥΊΘ§ΤδΟϊ≥Τ « Θ§ΗΟΦΛΥΊΒΡ÷ς“ΣΉς”Ο « ΓΘ
Θ®2Θ©¥”ΨΞΦβΚΆ…λ≥ΛΨΞ÷–Ης÷÷ΦΛΥΊΒΡΚ§ΝΩΩ¥Θ§…ζ≥ΛΥΊΚΆ≥ύΟΙΥΊ «÷ς“ΣΆ®Ιΐ¥ΌΫχœΗΑϊΒΡ Θ§¥”Εχ¥ΌΫχ÷≤ΈοΒΡ…ζ≥ΛΘΜœΗΑϊΖ÷Ν―ΥΊ÷ς“Σ «Ά®Ιΐ¥ΌΫχœΗΑϊΒΡ Θ§¥”Εχ¥ΌΫχ÷≤ΈοΒΡ…ζ≥ΛΓΘ
Θ®3Θ©Ά®ΙΐΫχ“Μ≤ΫΒΡ―–ΨΩΒΟ÷ΣΘ§œΗΑϊΖ÷Ν―ΥΊΩ…Ά®Ιΐ“÷÷Τ“Ε¬ΧΥΊΓΔΚΥΥαΚΆΒΑΑΉ÷ ΒΡΫΒΫβΘ§¥”Εχ“÷÷Τ“ΕΤ§ΥΞάœΓΘΈΣΝΥ÷ΛΟςœΗΑϊΖ÷Ν―ΥΊ”–―”ΜΚ“ΕΤ§ΥΞάœΒΡΉς”ΟΘ§ΩΤ―ßΦ“…ηΦΤΝΥ“‘œ¬ Β―ιΘ§«κΡψΆξ…ΤœύΙΊ≤Ϋ÷ηΘ§≤ΔΖ÷Έω Β―ιΫαΙϊΓΘ
ΔΌ Β―ι‘≠άμΘΚ“ρ“Ε¬ΧΥΊΒΡ÷πΫΞΫΒΫβΕχΒΦ÷¬“ΕΤ§ ß¬Χ±δΜΤ «“ΕΤ§ΥΞάœΒΡΉνœ‘÷χΧΊΒψΘ§“ρ¥ΥΘ§Ω…Ά®Ιΐ”Ο
ά¥¥Πάμ–¬œ ΒΡάκΧε“ΕΤ§Θ§Φ«¬Φ“ΕΤ§ ß¬Χ±δΜΤΥυ–ηΒΡ ±Φδά¥÷ΛΟςΓΘ
ΔΎ Β―ι≤Ϋ÷ηΘΚ
ΒΎ“Μ≤ΫΘʯûâ§÷÷÷≤ΈοΓΔ ΒΡ“ΕΤ§ΥφΜζΖ÷≥…ΝΫΉιΘ§Ζ÷±π±ξΦ«ΈΣΦΉΉιΓΔ““ΉιΓΘ
ΒΎΕΰ≤ΫΘΚ______________________________________Θ§‘Ύ““Ήι“ΕΤ§ΒΡœύΆ§ΈΜ÷ΟΆΩΒ»ΝΩΒΡ’τΝσΥ°ΓΘ
ΒΎ»ΐ≤ΫΘΚ ΦΉΓΔ““ΝΫΉι“ΕΤ§ ß¬Χ±δΜΤΥυ–ηΒΡ ±ΦδΓΘ
Δέ Β―ιΫαΙϊ‘Λ≤βΘΚ____________________________________________________ΓΘ
Δή Β―ιΫα¬έΘΚ________________________________________________________ΓΘ
Ρ≥Ά§―ßΑ¥œ¬±μΥυ Ψ÷Τ≥…ΝΌ ±ΉΑΤ§Ϋχ––”–ΙΊ Β―ιΓΘ
| Ήι±π | ≤ΡΝœ | Β―ιΧθΦΰ | Ιέ≤λΡΎ»ί |
| A | Ϋΰ≈ίΙΐΒΡΜ®…ζ÷÷Ή” | «εΥ°ΓΔΥ’ΒΛΔσ»Ψ“ΚΓΔ50%ΨΤΨΪ | œΗΑϊ÷–Ή≈…ΪΒΡ–ΓΩ≈ΝΘ |
| B | »ΥΒΡΩΎ«Μ…œΤΛœΗΑϊ | X»Ψ“Κ | œΏΝΘΧε |
| C | ≤Λ≤Υ“Ε | «εΥ° | “Ε¬ΧΧε |
| D | Ήœ…Ϊ―σ¥–ΝέΤ§“ΕΆβ±μΤΛ | 0.3g/mL’αΧ«»ή“Κ | ÷ ±ΎΖ÷άκ |
ΫΪ…œ ωΗςΉι Β―ι≤ΡΝœΑ¥±μ÷– Β―ιΧθΦΰΫχ––œύΙΊ¥ΠάμΚσΘ§÷Τ≥…ΝΌ ±ΉΑΤ§Θ§Ζ≈‘Ύœ‘ΈΔΨΒœ¬Ιέ≤λΓΘΖ÷ΈωΜΊ¥πΘΚ
Θ®1Θ©AΉι Β―ιΒΡΡΩΒΡ « Θ§50%ΨΤΨΪΒΡΉς”Ο «
Θ®2Θ©BΉι Β―ιΒΡX»Ψ“Κ « Θ§œ‘ΈΔΨΒœ¬Ω¥ΒΫΒΡœΏΝΘΧε≥ …ΪΓΘ
Θ®3Θ©‘ΎCΉι Β―ι÷–Θ§“Σ―Γ»Γ≤Λ≤Υ“Ε…‘¥χ–©“Ε»βΒΡœ¬±μΤΛΘ§‘≠“ρ «
Θ®4Θ©DΉι Β―ι÷–Θ§―ÔϹœ…Ϊ―σ¥–ΝέΤ§“ΕΒΡ‘≠“ρ « ΓΘ
(7Ζ÷Θ©ΒΆΈ¬Τ» Ι÷≤ΈοœΗΑϊ≤ζ…ζ¥σΝΩΕ‘œΗΑϊ”–ΚΠΒΡΙΐ―θΜ·ΈοΘ§»γ÷§÷ Ιΐ―θΜ·ΈοΘ®MDAΘ©ΓΘ
| Ήι±π | ¥Πάμ | ≈ύ―χΈ¬Ε» | SODΜν–‘[U/(gΓΛmin)] |
| 1 | ’τΝσΥ°ΫΫΙύ | 25Γφ | 7.3 |
| 2 | ΔΌ | ΔΎ | 9.4 |
| 3 | 0.5 nmol/LΥ°―νΥαΫΫΙύ | 5Γφ | 10.3 |
| 4 | 1.0 nmol/LΥ°―νΥαΫΫΙύ | 5Γφ | 11.6 |
| 5 | 1.5 nmol/LΥ°―νΥαΫΫΙύ | 5Γφ | 13.6 |
| 6 | 2.0 nmol/LΥ°―νΥαΫΫΙύ | 5Γφ | 8.5 |
| 7 | 2.5 nmol/LΥ°―νΥαΫΫΙύ | 5Γφ | 7.9 |
| 8 | 3.0 nmol/LΥ°―νΥαΫΫΙύ | 5Γφ | 6.5 |
Θ®1Θ©±μ÷–ΔΌ « Θ§ΔΎ « ΓΘ Β―ι…ηΦΤ ±ΟΩΉι»Γ50÷ξΥ°ΒΨ”ΉΟγΘ§Εχ≤Μ «1÷ξΘ§ΡΩΒΡ « ΓΘ
Θ®2Θ©±Ψ Β―ι–η“ΣΩΊ÷ΤΒΡΈόΙΊ±δΝΩ”– ΓΔ ΓΘ
Θ®3Θ©Ήι±π1ΚΆ2Ε‘’’Ω…ΒΟΒΡΫα¬έ «ΘΚ Θ§Υ°ΒΨ”ΉΟγΉ‘…μΩ…‘ω«ΩΩΙάδ–‘ΓΘ‘Ύ5ΓφΒΡΜΖΨ≥œ¬Θ§2.0 nmol/LΒΡΥ°―νΥαΕ‘Υ°ΒΨ”ΉΟγΩΙάδ–‘ΒΡ”Αœλ « Θ®ΧνΓΑ‘ω«ΩΓ±ΜρΓΑΦθ»θΓ±Θ©ΓΘ