��Ŀ����
8��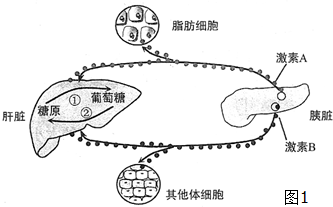 ��ͼ1��ʾ����Ѫ�ǵ��ڵĻ�����ʽ��ͼ�Т١��ڱ�ʾ�������̣�����A��B�����ڸ���ʱ�ֱ�ٽ����̢١��ڣ��ش��������⣨[]����ͼ����ĸ��������д���֣���
��ͼ1��ʾ����Ѫ�ǵ��ڵĻ�����ʽ��ͼ�Т١��ڱ�ʾ�������̣�����A��B�����ڸ���ʱ�ֱ�ٽ����̢١��ڣ��ش��������⣨[]����ͼ����ĸ��������д���֣�����1����ͼ����������A���ȸ�Ѫ���أ�B���ȵ��أ����ȵ�Bϸ�����ڣ���������ȱ������[B]���ɵ�������
��2����Ѫ�ǵ����У��뼤��A ����ͬ���õļ�����C��
A���Լ��� B��������
C���������� D���ټ�״����
��3��ͼ1�м�����ѪҺ�е�Ũ�Ⱦ��ܵͣ�������Ѫ���ȶ��ĵ�������ȴ�������ģ���˵�����صĵ������þ��и�Ч�ԣ�����A��֬��ϸ������ϸ���������ض����壬˵���������þ��������ԣ�
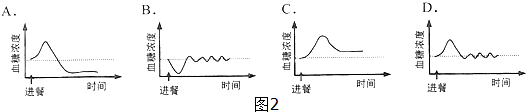
��4����ͼ2����ͼ�У��ɱ�ʾ�����˲ͺ�Ѫ��Ũ�ȱ仯���Ƶ�������D��
���� ��1���ȵ��ص����ã��ٴٽ�����֯��ϸ����Ѫ�ǵ����գ��ڴٽ������ǵ������ֽ⣻�۴ٽ����ࡢ������֯�ϳ���ԭ���ܴٽ�������ת��Ϊ�������ʣ������Ƹ���ԭ�ķֽ⣻�����Ʒ�������ת��Ϊ�����ǣ�
��2���ȸ�Ѫ���ص����ã��ٴٽ�����ԭ�ֽ⣻�ڴٽ���������ת��Ϊ�����ǣ�
��3���������ص����ã�����Ѫ�ǣ��ٽ����ȣ�
��� �⣺��1������A�����ڸ���ʱ�ٽ����̢٣�������ԭ�ֽ�������ǣ��ʼ���A���ȸ�Ѫ���أ�����B�����ڸ���ʱ�ٽ����̢ڣ��������Ǻϳɸ���ԭ���ʼ���B���ȵ��أ����ȵ�Bϸ�����ڣ���������ȱ������B�ȵ��أ��ɵ�������
��2����Ѫ�ǵ����У�����A�ȸ�Ѫ���ؾ�������Ѫ�ǵ����ã����뼤��A�ȸ�Ѫ��������ͬ���õļ�����C�������أ�
��3��ͼ1�м�����ѪҺ�е�Ũ�Ⱦ��ܵͣ�������Ѫ���ȶ��ĵ�������ȴ�������ģ���˵�����صĵ������þ��и�Ч�ԣ�����A��֬��ϸ������ϸ���������ض����壬˵���������þ��������ԣ�
��4���ͺ������ڸ������˴����������ǣ�Ѫ��Ũ�Ȼ����ߣ��������ȵ����ڵ��ȵ����ܵ����ǵĴ�л���ٽ�Ѫ�Ǻϳ���Ԫ������Ѫ�ǵķֽ⣬����Ѫ�ǵ�Ũ�ȣ����������˵�Ѫ��Ũ��ά����һ���ȶ���ˮƽ�ϣ���ѡ��D��
�ʴ�Ϊ��
��1���ȸ�Ѫ���� �ȵ��� �ȵ�B B
��2��C
��3����Ч ����
��4��D
���� ���⿼��Ѫ�ǵ��ڵ����֪ʶ��Ҫ����ʶ������Ѫ�ǵ��ڵľ�����̣������ȵ��غ��ȸ�Ѫ���صķ���ϸ�������幦�ܼ����ϵ��ͬʱ��������ϵʵ�ʣ�������ѧ��֪ʶ�������������е�����ѧ���⣮

| A�� | �ڶ����� | B�� | ������ĸϸ�� | C�� | ��ԭϸ�� | D�� | ������ĸϸ�� |
| A�� | ��÷ֹ�� | B�� | ϥ������ | C�� | ���ַ��� | D�� | ˱�̷��� |
| A�� | �³´�л����������ȫ������ѧ�仯���ܳ� | |
| B�� | ����ȿ��Խ���ͬ�����ã��ֿ��Խ����컯���� | |
| C�� | �������Զ����Ľ��в����³´�л�����ϳ���ǵ��� | |
| D�� | �������ĸ���Ԫ�غͻ��������³´�л�����ʻ��� |
| �Թܺ� | ||
| 1 | 2 | |
| PH=6.8�Ļ���Һ���Σ� | 20 | 20 |
| 1%������Һ���Σ� | 10 | - |
| 1%������Һ���Σ� | - | 10 |
| �Ʊ���Һ���Σ� | 5 | 5 |
| �Թܺ� | |||
| 1 | 2 | 3 | |
| PH=6.8�Ļ���Һ���Σ� | 20 | 20 | 20 |
| 1%������Һ���Σ� | 10 | 10 | 10 |
| ˮ�ܴ���5min | 37�� | ��ˮ | ��ԡ |
| �Ʊ���Һ���Σ� | 5 | 5 | 5 |
| 1 | 2 | 3 | 4 | |
| PH=6.8�Ļ���Һ���Σ� | 20 | 20 | 20 | 20 |
| 1%������Һ���Σ� | 10 | 10 | 10 | 10 |
| ����ˮ���Σ� | 10 | - | - | - |
| 9%Nacl���� | - | 10 | - | - |
| 1%CuSO4���� | - | - | 10 | - |
| 1%Na2SO4���� | - | - | - | 10 |
| �Ʊ���Һ���Σ� | 5 | 5 | 5 | 5 |
��1����1���ֵ�ʵ����Ϊ��֤������Һ���ۣ�ø����רһ�ԣ�
��2����1��2��3�о�����PH=6.8�Ļ���Һ����������ʹ��Ӧ���������˵�pH�����н��У���1����3��ʾʵ��Ӧ��37��ˮԡ���������½��У�
��3����1����2��ʾʵ���ڼ��ʵ����ʱ���ʺ��õ�ʵ���Լ��ֱ�������Լ�����Һ��
��4���ڱ�3��4֧�Թ��и��μ�1�ε�Һ���������¶��±���5min���Թ�2����ɫ��ʧ���ٹ�5min���Թ�1��4�е���ɫҲ��ʧ�����Թ�3һֱ����ɫ������ʵ����˵��Cl-��Cu2+ �ֱ�ٽ���������Һ����ø�Ļ��ԣ�
 ��ͼ�Ƕ���ϸ��ʾ��ͼ�����ͼ�ش�[]������ţ���
��ͼ�Ƕ���ϸ��ʾ��ͼ�����ͼ�ش�[]������ţ���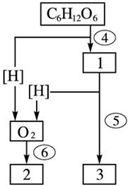 ͼ�������������̵�ͼ�⣮�����ͼ�ش��������⣮
ͼ�������������̵�ͼ�⣮�����ͼ�ش��������⣮