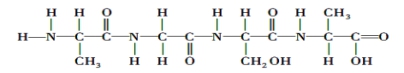
��Ŀ����
��֪�ݲ����������κ���������ʱ���йء�Ϊ�˲ⶨ��ͬ���������ݲ����������εĺ�����ijͬѧ�����һ��ʵ�飬ʵ����ϡ��Լ����þ߰������̶���Һ�ܡ���ɫ�ܡ���ͬŨ�ȵ��������Ʊ���Һ���������ε���ɫ������ͬ�����������ݲ���Һ�ȡ��ش�������⣺
(1)����������ʵ�鲽�衣
�ٱ��ܵ��Ʊ�����_______________________����ɫ���Ƴ���ɫ��dz��ͬ��ϵ�б��ܡ�
����Ʒ�ܵ��Ʊ����ÿ̶���Һ�ֱܷ���ȡһ������_____________________________���ӵ���ͬ�ı�ɫ���У�Ȼ���ڸ�����ɫ���м����������ɫ��������ɫ���õ���Ʒ�ܡ�
�۽�ÿ��_______�ֱ���ϵ�б��ܽ��бȽϣ��ҳ�����Ʒ����ɫ��dz_________�ı��ܣ��ù����������ƺ�����������Ʒ���е��������κ�������¼����Ʒ���������εĺ�����
(2)�ݲ����������в����ϸ����Ҫ��_______________��
(3)�ݲ˵�������������Ȼ�����ֳɵľ��֣�����Ϊ�˴ﵽĿ�Ļ���Ҫ��ʵ���������ض�������±���ɸѡ������ϸ�����������䷽����ش��������
KH2PO4 | Na2HPO4 | MgSO4��7H2O | FeCl3 | X | ά���� | ��֬ |
1.4 g | 2.1 g | 0.2 g | 0.1 g | 1 g | �� | 15 g |
�ٴ����������Ͽ�������������_______�����������гɷ�XΪĿ�ľ��ṩ____________���Ʊ�����������һ�����˳���Ǽ�����������ܻ��������____________��
����ͼA��B�Ǵ����������������ֽ��ַ�����C��D�ǽ��ֺ�������Ч����ijͬѧ������������ͼC��ʾЧ����������õĽ��ַ�����[����]______________________([����]ѡ�A����B��)������ǰ��Ҫ����������Ƿ���Ⱦ����ⷽ���ǽ�δ���ֵ��������������¶����������۲�____________��

 ����˼ά�żӿ���ϵ�д�
����˼ά�żӿ���ϵ�д� �����Ծ�ϵ�д�
�����Ծ�ϵ�д�

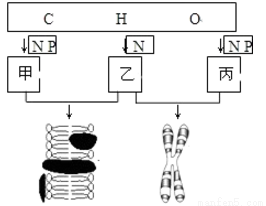
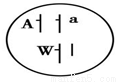

 �����
�����