��Ŀ����
7��ͼΪ�����Ŵ���������ΪA��a���������Ŵ���������ΪB��b���ļ�ϵͼ������һ���Ŵ�������λ�ڳ�Ⱦɫ���ϣ���һ��λ��XȾɫ���ϣ�����˵����ȷ���ǣ�������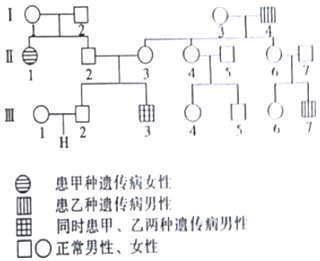
| A�� | �������������Ŵ����ķ����ʼ��Ŵ���ʽʱ�����ѡ���ڻ���ϵ�е��� | |
| B�� | �ס����Ŵ������Ŵ���ʽ����Ϊ��XȾɫ�������Ŵ�����Ⱦɫ�������Ŵ� | |
| C�� | ��-3�ļ����Ŵ������²�������Դ��I-1��I-2�������Ŵ������²�������Դ�ڢ�-4 | |
| D�� | ��-2�Ļ����ͼ�����ΪAAXBY��$\frac{1}{3}$��AaXBY��$\frac{2}{3}$ |
���� ���������ͼʾ������֪����-1�͢�-2���ײ�����������һ�����ײ���Ů����������������Ϊ���ԣ����Կ�Ů����Ů�������ǰ��ԡ���˵���ײ�Ϊ��Ⱦɫ�������Ŵ�������-2�͢�-3�����Ҳ�����������һ�����Ҳ��Ķ��ӣ�˵���ò�Ϊ�����Ŵ���������֪��һ���Ŵ�������λ�ڳ�Ⱦɫ���ϣ���һ��λ��XȾɫ���ϡ���˵���Ҳ�Ϊ��XȾɫ�������Ŵ�����
��� �⣺A�������Ŵ����ķ�����ʱ��Ӧ����Ⱥ�е��飬A����
B�����ݷ�����֪���ס����Ŵ������Ŵ���ʽ����Ϊ��Ⱦɫ�������Ŵ�����XȾɫ�������Ŵ���B����
C����-3�Ļ�����ΪaaXbY��������Ŵ������²�������Դ�ڢ�-1���-2����-3���-4���������Ŵ������²�������Դ�ڢ�-4��C����
D����-2�Ļ�������AaXBY����-3�Ļ�������AaXBXb�����Ԣ�-2�Ļ����ͼ�������$\frac{1}{3}$AAXBY��$\frac{2}{3}$AaXBY��D��ȷ��
��ѡ��D��
���� ������ϵ��ͼ�����������Ŵ�����Ҫ�������ռ��������Ŵ��������ͼ��ص㣬�ܸ���ϵ��ͼ�������Ϣ��ȷ�жϼס����Ŵ������Ŵ���ʽ����Ӧ����Ļ����ͣ������û���������϶��ɼ�����ظ��ʣ����ڿ��������Ӧ�ò�εĿ��飮

 ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д� ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д�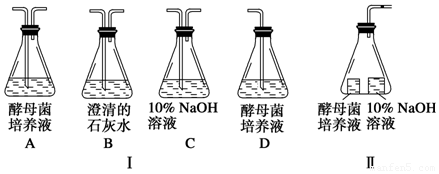
��1�������ʵ��Ŀ��ѡ��װ����ţ�������ʵ�����װҪ������װ�ÿ��ظ�ʹ�ã���
���������µ�װ����ţ�C��A��B����C��B��A��B�������������µ�װ����ţ�D��B��
��2��װ����Cƿ�������ǣ����տ�����CO2���ų����ʵ�����ĸ��ţ�Bƿ�г����ʯ��ˮ���������CO2�Ĵ��ڣ�
����ȤС������ͼ����ʾװ�ã���Ƥ���ϵ����Ϊ���к�ɫҺ�εĿ̶Ȳ����ܣ���̽����ĸ����ϸ���������ͣ�
��1����õ�ʵ����ۻ�����ͬʱ���ö���ʵ�飬���ʶ���ʵ��װ�ã������װ�ñ��Ϊ�������ƣ�װ�â���õ�����ˮ����NaOH��Һ�⣬���������װ�â���ͬ��
��2����Ԥ�����������ϵ�������д����
| ��� | װ���к�ɫҺ�ε��ƶ����� | ���� | |
| װ�â� | װ�â� | ||
| 1 | ������ | �ڲ��ƶ� | ֻ������������ |
| 2 | �۲��ƶ� | ������ | ֻ������������ |
| 3 | ������ | ������ | �Ƚ��������������ֽ����������� |
| A�� | �׳�ϸ�����������壬��ϸ��ֻ��ͨ��C6H12O6��2C3H6O3+�������� | |
| B�� | ����˾���ϸ���ʻ����в������� | |
| C�� | ������й�����õij�����Ҷ���� | |
| D�� | ��ĸ���ĸ߶����帺��ϳɵ����� |
| A�� | ϸ����������ֳ���� | B�� | ϸ��Ĥ�ϵ��ǵ����������� | ||
| C�� | ϸ��������ṹ�����仯 | D�� | ϸ��Ĥ�ϵ��ǵ������ʼ��� |
| A�� | �ݼ���ѭ������ | B�� | �����͵������� | ||
| C�� | �����������ݼ� | D�� | ������ѭ������ |
| A�� | ����ɫ��ʱ�����Թ��м������Һ��ʹҺ��߶ȵ�����Һϸ�� | |
| B�� | ʹ��Ѫ������������ĸ��ʱ��Ҫ�Ƚ�����Һ��Ͼ�����ȡ�� | |
| C�� | �����ƾ�ʱ������ֱ�ӽ��ظ������Һ���������Һ�۲���ɫ�仯 | |
| D�� | �۲���и�����˿����ʱ������֮��Ưϴ�Ϳ�������������ҺȾɫ |
| A�� | ������֭�е����Ǻ�ֲ��ϸ���е���ά�ض����ڶ��� | |
| B�� | ������ͬ���ǡ�֬���������������ֽ����ͷŵ���������ͬ�� | |
| C�� | ϸ�����ڵĺ���ֻ���������ǣ�ϸ�����еĺ���ֻ������ | |
| D�� | ���������е����ʡ���֬�ȣ��ܲ��뵰���ʺ�֬�ʵĺϳ� |
| A�� | ���������Էֲ����� | |
| B�� | û�зֲ����� | |
| C�� | ɭ�����зֲ�����ԭ��û�зֲ����� | |
| D�� | ֲ���зֲ�������û�зֲ����� |
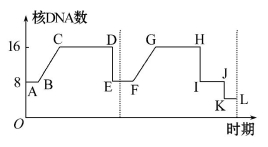
 ������Һ����������ʼ������һ��ϸ���к�DNA���ı仯����ͼ��ʾ������������ȷ���ǣ� ��
������Һ����������ʼ������һ��ϸ���к�DNA���ı仯����ͼ��ʾ������������ȷ���ǣ� ��