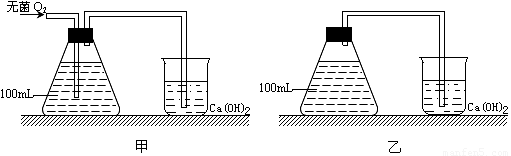
��Ŀ����
��֪��ĸ���ܽ�����������Ҳ�ܽ��������������ֽ�һ������ĸ������ʢ������������Һ����ƿ�У�����ƿ�ڽ��������������Ѿ�ѧ�������֪ʶ�ش��������⡣
��1���տ�ʼʱ����ĸ�����Уߣߣߣߣߣߣ�ԭ���ǣߣߣߣߣߣߣߣߣߣߣߣߣ�д���ù����к������õķ�Ӧʽ�ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
��2��һ��ʱ������ţߣߣߣߣߣ߽��ͣ���ĸͬʱ���Уߣߣߣߣߣߺͣߣߣߣߣߣߡ�
��3������ʱ������ӳ�����ĸֻ���Уߣߣߣߣߣߣ��ù��̺������õIJ���Ϊ�ߣߣߣߣߣߣߣߣߣߣߡ�
��4��������к������õ������������ù��̵ķ�ӦʽΪ�ߣߣߣߣߣߣߣߣߣߣߣߡ�
��1������������ƿ����һ��Ũ�ȵ�������C6H12O6+6H2O+6O2��6CO2+12H2O������
��2������Ũ�ȣ�������������������
��3�������������ƾ��Ͷ�����̼
��4��C6H12O6��2C3H6O3+����

��ϰ��ϵ�д�
 ������ĩ��ϰ��ѵ��ϵ�д�
������ĩ��ϰ��ѵ��ϵ�д� С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д�
�����Ŀ
 ��ѧ�������֪ʶ�ش��������⡣
��ѧ�������֪ʶ�ش��������⡣