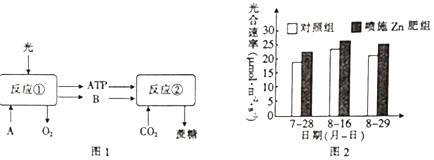
��Ŀ����
7����ͼ����̬ϵͳ��̼ѭ���ļ���ʾ��ͼ������ݻش��������⣺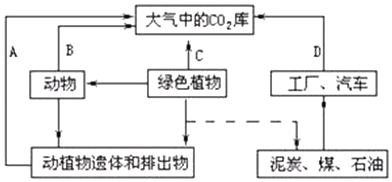
��1��ͼ��ȱ�ٵ�һ����Ҫ��ͷ�ǴӴ����е�CO2����ɫֲ�������CO2��������Ⱥ����ͨ������Ĺ��������ʵ�֣�̼Ԫ�ؽ�������Ⱥ�����Ⱥ���ڲ����л������ʽ���ݣ�
̼Ԫ��������Ⱥ���������֮���ѭ����ʽ��CO2��
��2��ͼ��B��C���̶�������Ⱥ���е�̼�ص������еĹ��̣�ʵ���϶���ͨ������ĺ���������ʵ�֣�
��3���������̬ϵͳ�ijɷ�����������A���̵��������ڷֽ��ߣ�A������ָ�ֽ����ã�
��4��������̼ѭ������������ú��ʯ�͵Ȼ�ʯȼ�ϵ�ȼ�գ���ΪD��;������һ���ţ�������CO2
�ĺ�������ɵ���Ҫ��������������ЧӦ��������ЧӦ�Ĵ�ʩ�У�ֲ�����֡���������Դ�������һ��ɣ�
���� ��̬ϵͳ��̼ѭ����
��̼�������������Զ�����̼��̼������ʽ���ڵģ�
��̼��������������Ⱥ��֮�����Զ�����̼��ʽ����ѭ���ģ�
��̼������Ⱥ���У��Ժ�̼�л�����ʽ���ڣ�
�ܴ����е�̼��Ҫͨ��ֲ�������ý�������Ⱥ�䣮
������Ⱥ���е�̼ͨ����ֲ��ĺ������á�����ķֽ����á���ʯȼ�ϵ�ȼ�յȷ�ʽ���Իص������У�
��ͼ������A��B��C��ʾ����Ⱥ��ĺ������ã�E��ʾ������ã�D��ʾȼ�գ�
��� �⣺��1��ͼ��ȱ�ٴӴ����е�CO2����ɫֲ��ļ�ͷ����ɫֲ��ͨ��������ý������е�CO2�̶�����ԭ���л��ﴢ��������̼Ԫ�ؽ�������Ⱥ�����Ⱥ���ڲ����л������ʽ���ݣ�̼Ԫ��������Ⱥ���������֮���ѭ����ʽ��CO2��
��2��ͼ��B��C���̶�������Ⱥ���е�̼�ص������еĹ��̣�ʵ���϶���ͨ������ĺ���������ʵ�֣�
��3���������̬ϵͳ�ijɷ�������A������ָ�ֽ��ߵķֽ����ã�����A���̵��������ڷֽ��ߣ�
��4��������̼ѭ������������ú��ʯ�͵Ȼ�ʯȼ�ϵ�ȼ�գ���ΪD������һ���ţ�������CO2�ĺ�������ɵ���Ҫ��������������ЧӦ��������ЧӦ�Ĵ�ʩ�У�ֲ�����֡���������Դ��
�ʴ�Ϊ��
��1���Ӵ����е�CO2 ����ɫֲ�� ������� �л��� CO2
��2����������
��3���ֽ��� �ֽ�����
��4��ú��ʯ�͵Ȼ�ʯȼ�ϵ�ȼ�գ���ΪD�� CO2 ����ЧӦ ֲ�����֡���������Դ
���� ���⿼����̬ϵͳ���ܣ��ۺϿ���������ѭ�������������Ĺ�ϵ�����ڿ��鿼����ʶͼ������������ѧ֪ʶҪ�㣬����֪ʶ��������ϵ���γ�֪ʶ����ṹ����������������ѧ֪ʶ��ȷ�ж���������������ڿ���ʶ�Ǻ������εĿ��飮

 ��У����ϵ�д�
��У����ϵ�д�| A�� | ��ø���ܺϳ�ˮ��ø���ڷֽ�˥�ϵ�ϸ���� | |
| B�� | �������ڸߵ�ֲ��ϸ����˿���ѵļ�����ɱ��� | |
| C�� | Ҷ�����������庬������ܵ�ɫ�� | |
| D�� | ֲ��ϸ���ʱڷ��븴ԭ������Һ����ϸ��Һ��Ũ�������� |
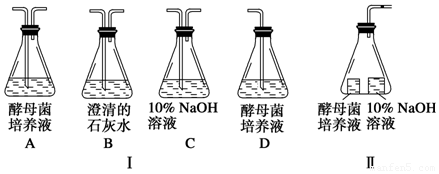
��1�������ʵ��Ŀ��ѡ��װ����ţ�������ʵ�����װҪ������װ�ÿ��ظ�ʹ�ã���
���������µ�װ����ţ�C��A��B����C��B��A��B�������������µ�װ����ţ�D��B��
��2��װ����Cƿ�������ǣ����տ�����CO2���ų����ʵ�����ĸ��ţ�Bƿ�г����ʯ��ˮ���������CO2�Ĵ��ڣ�
����ȤС������ͼ����ʾװ�ã���Ƥ���ϵ����Ϊ���к�ɫҺ�εĿ̶Ȳ����ܣ���̽����ĸ����ϸ���������ͣ�
��1����õ�ʵ����ۻ�����ͬʱ���ö���ʵ�飬���ʶ���ʵ��װ�ã������װ�ñ��Ϊ�������ƣ�װ�â���õ�����ˮ����NaOH��Һ�⣬���������װ�â���ͬ��
��2����Ԥ�����������ϵ�������д����
| ��� | װ���к�ɫҺ�ε��ƶ����� | ���� | |
| װ�â� | װ�â� | ||
| 1 | ������ | �ڲ��ƶ� | ֻ������������ |
| 2 | �۲��ƶ� | ������ | ֻ������������ |
| 3 | ������ | ������ | �Ƚ��������������ֽ����������� |
| A�� | �׳�ϸ�����������壬��ϸ��ֻ��ͨ��C6H12O6��2C3H6O3+�������� | |
| B�� | ����˾���ϸ���ʻ����в������� | |
| C�� | ������й�����õij�����Ҷ���� | |
| D�� | ��ĸ���ĸ߶����帺��ϳɵ����� |
| A�� | ������֭�е����Ǻ�ֲ��ϸ���е���ά�ض����ڶ��� | |
| B�� | ������ͬ���ǡ�֬���������������ֽ����ͷŵ���������ͬ�� | |
| C�� | ϸ�����ڵĺ���ֻ���������ǣ�ϸ�����еĺ���ֻ������ | |
| D�� | ���������е����ʡ���֬�ȣ��ܲ��뵰���ʺ�֬�ʵĺϳ� |
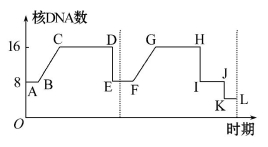
 ������Һ����������ʼ������һ��ϸ���к�DNA���ı仯����ͼ��ʾ������������ȷ���ǣ� ��
������Һ����������ʼ������һ��ϸ���к�DNA���ı仯����ͼ��ʾ������������ȷ���ǣ� ��