��Ŀ����
(12��)Ⱦ�϶��ȵ���ƣ�DCPIP��������̬������ɫ������ԭʱ�����ַۺ�ɫ����ɫ�����������ʵ�Ҷ����ƽ���ֳɼס���2�ݣ��ֱ�������������0.35mol/LNaCl��Һ��0.035mol/LNaCl��Һ��ҡ���Ƴ���Һ��Һ����Һ����������ʵ�飺
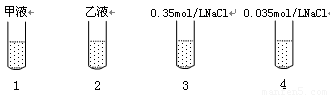
��ȡ�ɾ����Թ�4֧�����1��2��3��4�ţ��ںڰ�������ͼ��ʾ�ֱ���������������Һ���Լ���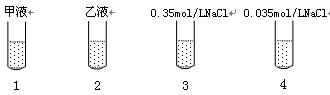
�ڽ���֧�Թ����ڹ���ǿ����ͬ�����˵Ĺ�Դ�£�����ÿ֧�Թ��м���2������ɫ�Ķ��ȵ������Һ��ҡ�ȣ��۲졣
��ʵ���������1���Թܲ������ݣ���Һ������ɫ��ɷۺ�ɫ�����ճ���ɫ��������֧�Թ������ݣ���Һ��������ɫ���ش����⣺
��1������ʵ������������
�� 1���Թ������е�����ɷ���  ��
��
�� 2���Թ�û�����������ԭ���� ��
�� 1���Թ�����ɫ��ʧ��ԭ���ǣ� ��
��2����ʵ���ʵ��ԭ����
��Ⱦ�϶��ȵ���ƣ�DCPIP��������̬������ɫ������ԭʱ�����ַۺ�ɫ����ɫ������Ϊ ��
�� ��
�� ��
��1����������O2��
�� 0.035mol/LNaCl��Ũ�ȵ���Ҷ������ʣ�0.035mol/LNaCl�ǵ�����Һ����Ҷ����ͨ����������ˮ���ѣ����ṹ���ƻ��������ܽ��й�����ã��ⷴӦ��û������������
�ۣ�������ã��ⷴӦ�ֽ�ˮ������ԭ�⣨[H]��NADPH������ԭ�ԭ���ȵ����ʹ�䣨������̬ת��ɻ�ԭ̬��������ɫ��
��2���ٹ�����ò�����ԭ����ָʾ����
��Ҷ����ֻ�б��ֽṹ�������Բ��ܽ��й�����ã�Ҷ�����ǹ�����õĻ�����λ����
���ڵ�������Ũ�ȣ���Һ�У������壩Ҷ���巢����ͨ������������ˮ�����ѣ���
����