��Ŀ����
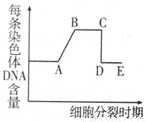
3����ͼΪT2�ɾ�����Ⱦϸ��ʵ��IJ��ֲ���ͽ�������ͼ�ش�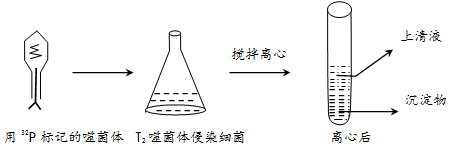
��1��T2�ɾ���Ļ�ѧ�ɷ���DNA�͵����ʣ��÷�����32P��ǵ���T2�ɾ����DNA��
��2��Ҫ���32P��ǵ��ɾ��壬�����ú�32P�Ĵ˾��������������ú�32P��������������ԭ���Dz���ֻ��Ӫ������������ڻ������ϸ���ڲ��ܷ�ֳ��
��3��ʵ������н����Ŀ����ʹ������ϸ���ϵ��ɾ�����ϸ�����룬���ĺ�����Խϸߵ��dz���������Һ�����������
��4����������ʱ���������������Һ�з�������ǿ������ܵ�ԭ���DZ���ʱ�������ϸ���ѽ⣬�ͷų����Ӵ��ɾ������ĺ��������Һ�У�
���� 1���ɾ���Ľṹ����������ǣ�C��H��O��N��S��+DNA��C��H��O��N��P����
2���ɾ��己ֳ���̣�������ע�루ע���ɾ����DNA�����ϳɣ������ߣ��ɾ����DNA��ԭ�ϣ�ϸ���Ļ�ѧ�ɷ֣�����װ���ͷţ�
3��T2�ɾ�����Ⱦϸ����ʵ�鲽�裺�ֱ���35S��32P����ɾ�����ɾ�����˾�����������ɾ�����Ⱦδ����ǵ�ϸ�����ڽ������н��裬Ȼ�����ģ��������Һ�ͳ������еķ��������ʣ�
��� �⣺��1��T2�ɾ���Ļ�ѧ�ɷ��ǵ����ʺ�DNA��������32P��ǵ���T2�ɾ����DNA��
��2���ɾ�����ϸ�����������ܶ���������������ڻ�ϸ���У����Ҫ���32P��ǵ��ɾ��壬�����ú�32P�Ĵ˾��������������ú�32P��������������
��3��ʵ������н����Ŀ����ʹϸ�������T2�ɾ��������ϸ�����룻32P��ǵ����ɾ����DNA���ɾ�����Ⱦϸ��ʱֻ��DNA����ϸ����������ϸ�����ĵ��������ʣ�������ĺ�����Խϸߵ��dz����
��4�������ɾ��������ʱ���������������Ⱦ��ϸ���ѽ��ͷų���32P��ǵ��Ӵ��ɾ��壬���ĺ�ֲ�������Һ�У���������Һ��������ǿ��
�ʴ�Ϊ��
��1��DNA�͵����� DNA
��2������ֻ��Ӫ������������ڻ������ϸ���ڲ��ܷ�ֳ
��3��ʹ������ϸ���ϵ��ɾ�����ϸ������ ������
��4������ʱ�������ϸ���ѽ⣬�ͷų����Ӵ��ɾ������ĺ��������Һ��
���� ������ͼ�⣬�����ɾ�����Ⱦϸ��ʵ�飬���ڴ������⣬��Ҫ����ע���ϸ�ڽ϶࣬��ʵ���ԭ����ʵ����õķ�����ʵ�������۵ȣ���Ҫ������ƽʱ��ѧϰ������ע����ۣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��������Ļ�����λ�Ǹ��� | |
| B�� | ��������ı����ǻ���Ƶ�ʵĶ���ı� | |
| C�� | ʨ�����Dz��鶯��������� | |
| D�� | ͬ���������Ⱥ��С�������С�ʷ��� |
| A�� | ��״�������������ֲ����ϸ���ӽ����� | |
| B�� | ͬһ������������״�������ͬ�Ļ����� | |
| C�� | �����˹����ӷ�ֳ�Ӵ�����ֳ��ʽ����������ֳ | |
| D�� | �˹������ܹ��˷�ԶԵ�ӽ������ϰ������� |
| A�� | S�;��ĵ�����ʮR�;� ע��С�� | |
| B�� | S�;���DNAʮR�;� ע��С�� | |
| C�� | R�;��ĵ�����ʮS�;� ע��С�� | |
| D�� | R�;���DNAʮS�;� ע��С�� |
| A�� | ����ͨ�� | B�� | ��������Ŀ��ٷ�ֳ | ||
| C�� | ��ֹ����ճ��Ӱ��۲� | D�� | ��ֹ�������ڵ���ȴˮ���������� |
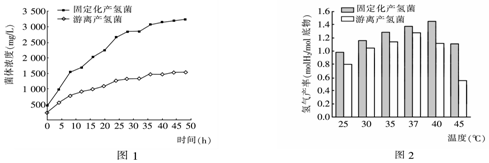
��1����ʵ���У��̶�����������õķ�����Ϊ����
��2����ͼ1��֪��������IJ������ȣ��̶����IJ�������������Ũ�����ӽϿ죬����Ҫԭ���������������γ������Ļ��������ڲ������������ֳ��
��3����ͼ2��֪���̶���������������������Ʊ������������ʸߡ����¶ȱ仯��Ӧ�Ը�ǿ��
��4��ʵ����ʹ�õķ�������Һ�ɷ������
| ����Һ�ɷ� | ������ | ������ | ţ��� | ��ĸ֭ | NaCl | K2HPO4 | ���װ��� | ��Ԫ�� |
| ������g/L�� | 20 | 4 | 2 | 1 | 2 | 1.5 | 0.5 | ���� |
��5��������ʵ���У�Ϊ�˳�ַ��ӹ̶�������������ƣ�����̽���̶��������������������������⣬����̽���̶�����������ظ�ʹ�õĴ�����
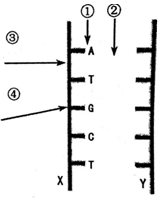 ��ͼ��һ�� DNA ���ӵĽṹʾ��ͼ�����ͼ�ش�
��ͼ��һ�� DNA ���ӵĽṹʾ��ͼ�����ͼ�ش�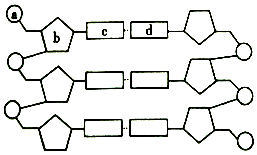 ��ͼ��ij����DNA���ӽṹƬ��ʾ��ͼ�����ͼ�ش����⣺
��ͼ��ij����DNA���ӽṹƬ��ʾ��ͼ�����ͼ�ش����⣺