��Ŀ����
Ϊ���о���Ӭ��ɫ�ͳ��ε��Ŵ����ɣ����й����������۾��ᡢ���۾��ᡢ���۳��ᣨҰ���ͣ�������ͬƷϵ�Ĺ�ӬΪ���ϣ������ӽ�ʵ�飬������¡�������ش�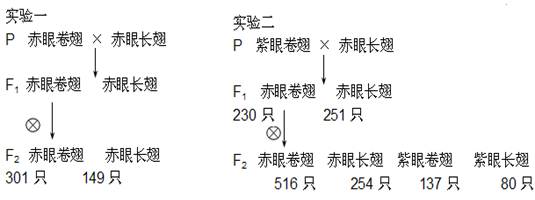
��1����������������ʵ��һ��ʵ���F1��������У����۱����ӽ�1:1��˵�����Ƴ��εĻ���λ�� Ⱦɫ���ϣ����ƹ�Ӭ��ɫ�ͳ��ε�2�Ի���Ĵ��ݷ��� ���ɡ�
��2���о����ڽ������Ϲ�Ӭ���ε��Ŵ�����ʱ�������������������ЧӦ���������ת����ʵ�������֤��
����ȡ����Ʒϵ����DNA������DNA�͵����ʵȳɷ��ڲ�ͬŨ�ȵ�NaCl��Һ���ܽ�Ȳ�ͬ��ʹDNA���������ʷ��룻Ϊ�˽�һ���ᴿ������DNA������95%����ƾ�������DNA����Һ����ýϴ�����DNA��Ʒ��Ӧ��PCR������������������DNAƬ�Ρ�
������͵��������������Ƭ���벡�����飬������۳���Ʒϵ��Ӭ���ܾ��ѡ����в����������� �����ܾ�����Ϊ����ϸ������Ϊ ��
�����������ܾ������ɳ棬ѡ��ɹ�����Ŀ�Ļ���Ĵ��۹�Ӭ���䡣�����ϼ�����������������볤��ı���Ϊ________��
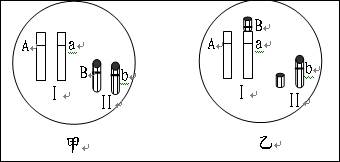
��3����һЩ�о����ڽ������Ϲ�Ӭ���ε��Ŵ�����ʱ��������۾���Ʒϵ�ͳ��۾���Ʒϵ��Ӭ�ھ������B������Ⱦɫ���ϴ���������������d�����û���������
�ٲ�����Ⱦɫ�彻�滥����d����Ĵ��� ���ܣ����ܣ�����F2�����볤��������ȡ�
���ڽ�һ���о��У��о�������������۾���Ʒϵ�ͳ��۾���
Ʒϵ������������ͬ���ֱ���d1��d2 ��ʾ����������Ⱦɫ���ϵ�λ������ͼ��ʾ��
����d1d1��d2d2������d1d2��������d1��d2 �����ڣ������ڣ���λ���������� ��
�������ϼ�������������۾���Ʒϵ�ͳ��۾���Ʒϵ�ӽ���
��������볤��ı���Ϊ________��
��1���� �������
��2������Ϊ�������壬Я��Ŀ�Ļ��� �ܾ��ѵ�ȫ����ǿ ��2:1
��3������ �ڲ����� d1d2û��λ��ͬԴȾɫ�����ͬλ���ϣ�2�֣� �� 3:1��2�֣�
���������������1����ʵ��һF1���۾����Ӭ�Խ�����м��о������г��ᣬ���Գ���Ϊ�����Ŵ�����������������ʵ��һ��ʵ���F1��������У����۱����ӽ�1��1��˵�����Ƴ��εĻ���λ�ڳ�Ⱦɫ���ϣ���ʵ���P���۾�������۳��ᣬF1���۾�����۳���=1��1��F1���۾����Ӭ�Խ���������۾�����۳�����۾�����۳���=6��3��2��1�����ܾ������������Դ�������ЧӦ��
��2����������Ϊ���̵����壬Я��Ŀ�Ļ������ܾ�����Ϊ����ϸ������Ϊ�ܾ��ѵ�ȫ����ǿ���Ӻ����ӽ���������������ľ����볤��ı���Ϊ2��1��
��3���ٵ�λ�����Ǵ�����ͬԴȾɫ����ͬλ�ÿ��������״�Ļ���ͼ���֪��d1d2û��λ��ͬԴȾɫ�����ͬλ�ã��������Dz����ڵ�λ�������������������ͼ������dz��͵Ļ��������ۣ�����Ʒϵ��Bbd1���ͣ����ۣ�����Ʒϵ��Bbd2���ӽ�������B��d������ͬһ��Ⱦɫ���ϣ����Ŵ���ѭ����ķ��붨�ɣ����ԣ����ۣ�����Ʒϵ��Bbd1��������������Bd1��b=1��1�������ۣ�����Ʒϵ��Bbd2��������������Bd2��b=1��1�����������������������ᣨBBd1d2��Bbd1��Bbd2���볤�ᣨbb���ı���Ϊ3��1��
���㣺���⿼�����ķ��붨�ɺ�������϶��ɡ����̵����֪ʶ�����ڿ���ѧ����������ѧ֪ʶ��۵㣬ͨ���Ƚϡ��������ۺϵȷ�����ijЩ����ѧ������н��͡������������������жϻ�ó���ȷ�Ľ��ۡ�

Ϊ̽��Ӱ��������ǿ�ȵ����أ�ijʵ��С�齫һ��Ũ�ȵ�С�������ʢ����������Һ����֧�Թ��У�1���Թܺ�2���ԹܵĶ�����̼Ũ�ȷֱ�����Ϊ0.05%��0.03%���׳����Ϊ��Դ���ƶ�̨�Ƹı��Դ���Թܵľ������ʵ�顣����ʵ�������Ƴ�ͼ������1������2.������ش�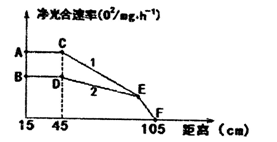
��1���ⶨ��������ʳ��˲ⶨ�����ͷ����⣬�����Բⶨ_____________��
��2����ͼ��֪������D�㾻������ʵ���Ҫ������_____________������EF�ξ�������ʵ���Ҫ������_____________��F��ĺ�����_____________��
��3����С��������̽��С�������������¶ȵ�Ԥʵ�飬ʵ�������±�����������̼Ũ��Ϊ0.05%�������ʵ���������������̽��ʵ��˼·��
| �¶ȣ��棩 | 10 | 20 | 30 | 40 | 50 |
| �������ͷ����������ʣ�mg��h-1�� | 9.67 | 14.67 | 19.67 | 21.67 | 19.33 |
��ʵ���л���Ӧ���ƺõ�������Ҫ�ر�����_____________��
Ԥʵ�����¶�Ϊ_______��ʱ������������ʱ��Ҷ������ADP���ƶ�������_____________��