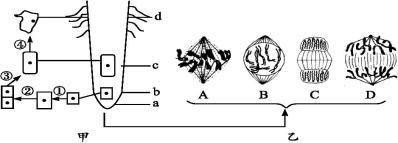
��Ŀ����
9�� ��ͼ��ʾ����������Ũ�ȶԽ�ĸ��ϸ����CO2������Ӱ�죬���ͼ�ش�
��ͼ��ʾ����������Ũ�ȶԽ�ĸ��ϸ����CO2������Ӱ�죬���ͼ�ش���1��A���ʾ��ĸ����֯�ͷŵ�CO2�϶࣬��ЩCO2��ϸ���ʻ��ʵIJ����ģ�ϸ���ṹ����
��2����ͥ��Ƶľ���������̣��Ƚ������죬����ȴ��30��ʱ��������ˮ��һ�����Ľ�ĸ �����֣��������Ⱥ�����һ��̳�ڲ���������һ�����ӸǺ������ʵ��ĵط����£�28�棩�������м���һ������Ŀ���������������������ĺ�������֤��ĸ����һ��ʼʱ���������������Ӷ������ڽ�ĸ�����������ӣ�
��3���ڼ�ͥ��ƹ����У�����������ˮ����Щˮ����Ҫ��Դ������������
��4�����������У���ĸ�������е����ִ�л�����ĵ����ʵ�����Ӫ�����ʣ������ǣ�������������������֮��Ϊ������������������=3��1��
��5��д����ĸ���������������ķ�Ӧʽ��C6H12O6+6H2O+6O2$\stackrel{ø}{��}$6CO2+12H2O+������
���� 1������ͼ�⣺��ͼ��A��������Ũ��Ϊ0��ֲ���ܽ��������������ͷų�������̼��
����A��B���������ӣ����������ܵ����ƣ�����CO2���ͷ���������٣�
����B��C���������������ӣ�ֲ�������������ǿ��CO2�ͷ������࣬����CO2���ͷ����ֲ������ӣ�
2��������������������ǽ�ĸ�������³´�л����Ϊ�������������ͣ�����������ԭ����
��1�������������£���Ӧʽ���£�C6H12O6+6H2O+6O2$\stackrel{ø}{��}$6CO2+12H2O+������
��2�������������£���Ӧʽ���£�C6H12O6$\stackrel{ø}{��}$2CO2+2C2H5OH+������
��� �⣺��1��ͼ��A��������Ũ�Ƚӽ�0����ĸ��ֻ�ܽ��������������ͷų�������̼�������������ij���Ϊϸ���ʻ��ʣ�
��2������ƾ�ʱ���ȼ����ĸ������Ҫ��ĸ���ȴ����������ͷ�ֳ��������Ŵ���м���һ��������Ϊ�˴洢һ���ֿ������ý�ĸ���Ƚ��������������������ӽ�ĸ����������������������������ͣ������ƾ���
��3����ƹ���������������ˮ������ƣ�ԭ���ǽ�ĸ���Ƚ������������ֽ����࣬����ˮ��������������������ƾ���
��4�����������У���ĸ�������������ܷ�ӦʽΪ��C6H12O6+6H2O+6O2$\stackrel{ø}{��}$6CO2+12H2O+�����������������ܷ�ӦʽΪ��C6H12O6$\stackrel{ø}{��}$2CO2+2C2H5OH+����������ĸ���������ִ�л�����ĵ����ʵ����������ǣ�������������������֮��Ϊ3��1��
��5�������������£���ĸ������������������Ӧʽ���£�C6H12O6+6H2O+6O2$\stackrel{ø}{��}$6CO2+12H2O+������
�ʴ�Ϊ��
��1��ϸ���ʻ���
��2�������������� ��������
��3����������
��4��������������������=3��1
��5��C6H12O6+6H2O+6O2$\stackrel{ø}{��}$6CO2+12H2O+����
���� ���⿼����ƺ���������Ҫ����ʶ�Dz�����ƺ������������P���л���ͣ����չ��ƺ���������ԭ������������������ϵʵ�ʣ������ѧ��֪ʶ�������������е�����ѧ���⣮

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ���á����̾��������ȵ��� | B�� | �������ﷴӦ�� | ||
| C�� | �����ʹ��� | D�� | �ӽ����� |
| A�� | ���κ���ʱ��Ӧ��������Ⱥ�����ﵽ$\frac{K}{2}$֮ǰ���в�ɱ | |
| B�� | ������Ȼ������������߱���������ϡ������Ⱥ��Kֵ | |
| C�� | ����������ṹ����Ⱥ���бȽϿ�֪�ȶ��͵ij�������� | |
| D�� | �������ռ���ɱ������۳�������Խ��ͺ������Ⱥ�ܶ� |
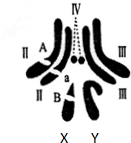
| ������� | ������� | ���� | ���� | |
| ��Ӭ | $\frac{3}{8}$ | 0 | $\frac{1}{8}$ | 0 |
| ��Ӭ | $\frac{3}{16}$ | $\frac{3}{16}$ | $\frac{1}{16}$ | $\frac{1}{16}$ |
��2���ù�Ӭ�롰��һ�ױ����ӽ����Ӵ��ij�����۴ƹ�Ӭ���Ӻ���ռ�ı���Ϊ$\frac{5}{6}$��
��3����ֻ�����Ӵ���Ӭ����ɫ�����ж����Ա���ѡ���ױ���Ӭ����ɫ������Ӧ��XbXb��XBY�����ù�Ӭ��A��B�������˻���������Ⱦɫ��ṹ���죮
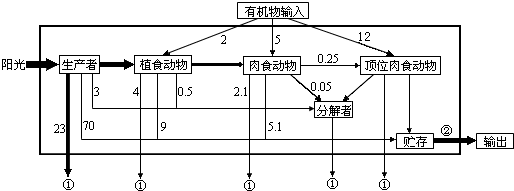
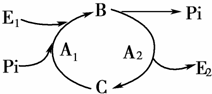
 ͼΪ������ϸ�����ڻ���֮�����ʽ�����ʾ��ͼ����ͼ������
ͼΪ������ϸ�����ڻ���֮�����ʽ�����ʾ��ͼ����ͼ������