0 140345 140353 140359 140363 140369 140371 140375 140381 140383 140389 140395 140399 140401 140405 140411 140413 140419 140423 140425 140429 140431 140435 140437 140439 140440 140441 140443 140444 140445 140447 140449 140453 140455 140459 140461 140465 140471 140473 140479 140483 140485 140489 140495 140501 140503 140509 140513 140515 140521 140525 140531 140539 203614
 ________。
________。
 +2I-+2H+===AsO
+2I-+2H+===AsO +I2+H2O,在碱性条件下发生的反应为AsO
+I2+H2O,在碱性条件下发生的反应为AsO
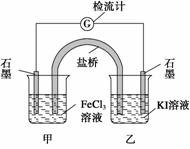

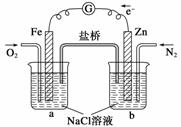
 ②
②