��Ŀ����
��10�֣� Ԫ�����ڱ��еڢ�A��Ԫ�صĵ��ʼ��仯�������;�㷺��
��1������Ԫ��ͬ��Ķ�����Ԫ�ص�ԭ�ӽṹʾ��ͼΪ________��
��2������Ϊ�ȡ��塢��Ԫ�طǽ�����(ԭ�ӵõ�������)�ݱ���ɵ��ж�������________(�����)��
a��Cl2��Br2��I2���۵� b��Cl2��Br2��I2��������
c��HCl��HBr��HI�����ȶ��� d��HCl��HBr��HI������
��3����ҵ�ϣ�ͨ������ת�����Ƶ�KClO3���壺

����ɢ��з�Ӧ���ܻ�ѧ����ʽ��
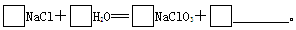
�ڢ���ת���Ļ�����Ӧ������________________���÷�Ӧ����������KClO3���������������������ԭ����____________________________________��
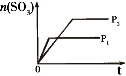
��4��һ�������£���ˮ��Һ��1 mol Cl����ClO(x��1��2��3��4)������(kJ)��Դ�С����ͼ��ʾ��
��D��________(�����ӷ���)��
��B��A��C��Ӧ���Ȼ�ѧ����ʽΪ________________(�����ӷ��ű�ʾ)��

��1��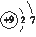 ��;��2��b��c��;
��;��2��b��c��;
��3�� 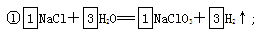 �ڸ��ֽⷴӦ �����£��������ˮ�е��ܽ������С����������;
�ڸ��ֽⷴӦ �����£��������ˮ�е��ܽ������С����������;
��4�� ��ClO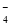 ; �� 3ClO��(aq)===ClO
; �� 3ClO��(aq)===ClO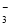 (aq)��2Cl��(aq)����H����117 kJ��mol��1
(aq)��2Cl��(aq)����H����117 kJ��mol��1
��������
�����������1������Ԫ��ͬ��Ķ�����Ԫ��F��ԭ�ӽṹʾ��ͼΪ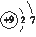 ;��2�� a��Cl2��Br2��I2���۵�ĸߵ�����Ӽ��������Ĵ�С�йأ�����Ԫ�صķǽ����Ե�ǿ���أ�����b��Ԫ�صķǽ�����Խǿ���䵥�ʵ������Ծ�Խǿ�����Cl2��Br2��I2����������Ԫ�صķǽ�����ǿ���йأ���ȷ��c��Ԫ�صķǽ�����Խǿ������Ӧ���⻯����ȶ��Ծ�Խǿ�����ȶ��ԣ�HCl>HBr>HI,���Ԫ�صķǽ����ԣ�Cl>Br>I����ȷ��d���������еĻ�ѧ��Խ������ѧ����Խ�����룬���иû�ѧ�������ʵ����Ծ�Խǿ������Ԫ�صķǽ�����ǿ���أ�����3�� ����ɢ��з�Ӧ���ܻ�ѧ����ʽΪ��
;��2�� a��Cl2��Br2��I2���۵�ĸߵ�����Ӽ��������Ĵ�С�йأ�����Ԫ�صķǽ����Ե�ǿ���أ�����b��Ԫ�صķǽ�����Խǿ���䵥�ʵ������Ծ�Խǿ�����Cl2��Br2��I2����������Ԫ�صķǽ�����ǿ���йأ���ȷ��c��Ԫ�صķǽ�����Խǿ������Ӧ���⻯����ȶ��Ծ�Խǿ�����ȶ��ԣ�HCl>HBr>HI,���Ԫ�صķǽ����ԣ�Cl>Br>I����ȷ��d���������еĻ�ѧ��Խ������ѧ����Խ�����룬���иû�ѧ�������ʵ����Ծ�Խǿ������Ԫ�صķǽ�����ǿ���أ�����3�� ����ɢ��з�Ӧ���ܻ�ѧ����ʽΪ��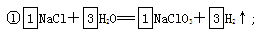 �ڴ�����֮���ת����ϵ��֪������ת���Ļ�����Ӧ�����Ǹ��ֽⷴӦ���÷�Ӧ����������KClO3���������������������ԭ���������£��������ˮ�е��ܽ������С���������壻��4������D��Ԫ�صĻ��ϼ�Ϊ+7�ۣ����Ժ���ClԪ�ص�+7�۵�������ClO
�ڴ�����֮���ת����ϵ��֪������ת���Ļ�����Ӧ�����Ǹ��ֽⷴӦ���÷�Ӧ����������KClO3���������������������ԭ���������£��������ˮ�е��ܽ������С���������壻��4������D��Ԫ�صĻ��ϼ�Ϊ+7�ۣ����Ժ���ClԪ�ص�+7�۵�������ClO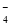 ;�ڸ���ͼʾ��֪��B��A��C��Ӧ���Ȼ�ѧ����ʽΪ3ClO��(aq)===ClO
;�ڸ���ͼʾ��֪��B��A��C��Ӧ���Ȼ�ѧ����ʽΪ3ClO��(aq)===ClO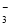 (aq)��2Cl��(aq)����H����117 kJ/mol.
(aq)��2Cl��(aq)����H����117 kJ/mol.
���㣺����ԭ�ӽṹʾ��ͼ��ʾ��Ԫ�صķǽ�����ǿ���Ƚϡ���ѧ��Ӧ���͵��жϡ���ѧ����ʽ���Ȼ�ѧ����ʽ����д��֪ʶ��

����ʵ�鷽���У����ܴﵽʵ��Ŀ�ĵ���(����)
ѡ�� | ʵ��Ŀ�� | ʵ�鷽�� |
A | ����CH3CH2Br��NaOH��Һ���Ƿ���ˮ�� | ��CH3CH2Br��NaOH��Һ���ȡ���ȴ��ȡ���ϲ�ˮ��Һ����ϡHNO3�ữ������AgNO3��Һ���۲��Ƿ��������ɫ���� |
B | ����Fe(NO3)2�����Ƿ����������� | ��Fe(NO3)2��Ʒ����ϡH2SO4�μ�KSCN��Һ���۲���Һ�Ƿ��� |
C | ��֤Br2��������ǿ��I2 | ��������ˮ����KI��Һ�У��ټ���CCl4�������ã��ɹ۲쵽�²�Һ�����ɫ |
D | ��֤Fe(OH)3���ܽ��С��Mg(OH)2 | ��FeCl3��Һ����Mg(OH)2����Һ�У����ɹ۲쵽�����ɰ�ɫ��Ϊ���ɫ |
�����о�Ŀ�ĺ�ʾ��ͼ�������( )
| A | B | C | D |
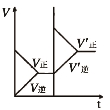
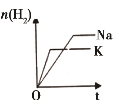
�� �� Ŀ �� | �̶��ݻ����ܱ������У�ѹǿ�Է�Ӧ�� 2SO2(g)+O2(g) | �̶��ݻ����ܱ������У��¶ȶԷ�Ӧ�� N2(g) +3H2(g) | �̶��ݻ����ܱ������У�����CO2Ũ�ȣ����Ѵ�ƽ��ķ�Ӧ�� CO(g)+H2O(g) | ������Na��K �ֱ�������ˮ��Ӧ |
ʾ �� ͼ |
|
|
|
|
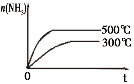
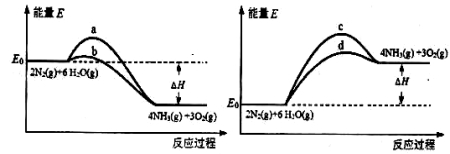
 2NH3
2NH3 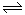 4NH3(g)+3O2(g) ��H��Q ��
4NH3(g)+3O2(g) ��H��Q ��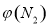 �뷴Ӧ�¶�T�Ĺ�ϵ��������ͼ��ʾ����������Ӧ��Q________0�����������������=������
�뷴Ӧ�¶�T�Ĺ�ϵ��������ͼ��ʾ����������Ӧ��Q________0�����������������=������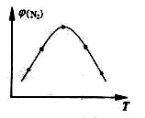
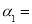 ______________��
______________��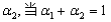 ʱ������ʼʱNH3�����ʵ���
ʱ������ʼʱNH3�����ʵ��� _________mol��
_________mol��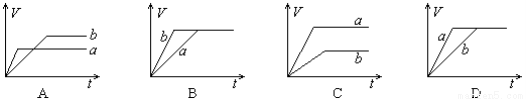 ��
��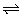 2SO3(g)��Ӱ��
2SO3(g)��Ӱ��