��Ŀ����
8������0.001mol•L-1����KMn04��Һ��δ֪Ũ�ȵ���ɫNa2SO3��Һ����Ӧ���ӷ���ʽ��2MnO4-+5SO32-+6H+=2Mn2++5SO42-+3H20�ش��������⣺
��1���õζ�ʵ�����������������е�ABDEFGH��
A����ʽ�ζ��ܣ�50mL�� B����ʽ�ζ��ܣ�50mL�� C����Ͳ��10mL�� D����ƿ E������̨ F���ζ��ܼ� G���ձ� H����ֽ I����ͷ�ι� J��©��
��2�����ü�ᡢ�ʽ�ζ���ʢ�����Ը��������Һ���Է���ԭ���������ܸ�ʴ�ܣ�
��3��ѡ����ָʾ����˵�����ɲ���Ҫ����Ϊ�ζ��յ�ʱ����Һ����ɫ��Ϊ�Ϻ�ɫ����
��4���ζ�ǰƽ������KMnO4��ҺҺ�棬�̶�ΪamL���ζ�����Һ��̶�ΪbmL����b-a��mL��ʵ������KMnO4��Һ����٣��ࡢ�٣������ݣ�b-a��mL����õ��Ĵ���Ũ�ȣ���ʵ��Ũ��С����С����
���� ��1�����Ը�����ؾ���ǿ�����ԣ�ʵ��ʱӦ����ʽ�ζ��ܣ���ɫNa2SO3��Һ�Լ��ԣ��ü�ʽ�ζ��ܣ��ζ����̻���Ҫ�ձ�����ƿ����ֽ���ζ��ܼк�����̨��
��2��������ؾ���ǿ�����ԣ��ܸ�ʴ�ܣ�
��3�����������Һ��ɫΪ�Ϻ�ɫ���ζ��յ�ʱ����Һ����ɫ��Ϊ��ɫ��
��4���ζ�����Һ�棬����ƫС�Լ�����ԭ��������c�����⣩=$\frac{c����ע����V������}{V�����⣩}$��������������V��������Ӱ�죬�Դ��ж�Ũ�ȵ���
��� �⣺��1�����Ը�����ؾ���ǿ�����ԣ�ʵ��ʱӦ����ʽ�ζ��ܣ���ɫNa2SO3��Һ�Լ��ԣ��ü�ʽ�ζ��ܣ��ζ����̻���ҪҪ�ձ�����ƿ����ֽ���ζ��ܼк�����̨������Ҫ�õ����ǣ���Ͳ����ͷ�ιܡ�©����
�ʴ�Ϊ��ABDEFGH��
��2��������ؾ���ǿ�����ԣ��ܸ�ʴ�ܣ�Ӧ����ʽ�ζ�����ȡ�������ü�ʽ��
�ʴ�Ϊ�����������ܸ�ʴ�ܣ�
��3�����������Һ��ɫΪ�Ϻ�ɫ���ζ��յ�ʱ����Һ����ɫ��Ϊ�Ϻ�ɫ������ָʾ��Ҳ�ܹ��۲�ζ��յ㣻
�ʴ�Ϊ������Ҫ����Ϊ�ζ��յ�ʱ����Һ����ɫ��Ϊ�Ϻ�ɫ��
��4���ζ�ǰƽ������KMnO4��ҺҺ�棬�̶�ΪamL���ζ�����Һ��̶�ΪbmL������ƫС����b-a��mL��ʵ������KMnO4��Һ����٣����ݣ�b-a��mL����õ��Ĵ���Ũ�ȣ����V������ƫС������c�����⣩=$\frac{c����ע����V������}{V�����⣩}$��������֪c�����⣩ƫС��
�ʴ�Ϊ���٣�С��
���� ������Ҫ�����˵ζ�������������ʹ���Լ��ζ����㣬�ѶȲ������������к͵ζ���������ԭ�ζ���ԭ���ǽ���ؼ���

��1���ڶ������У�Ԫ�صĵ�һ�����ܴ���B��N֮���Ԫ����3�֣�
��2��ijԪ��λ�ڵ������ڢ��壬���̬ԭ�ӵ�δ�ɶԵ��������̬̼ԭ�ӵ�δ�ɶԵ�������ͬ�������̬ԭ�ӵļ۲�����Ų�ʽΪ3d84s2��
��3����ϩͪ��CH2=C=O����һ����Ҫ���л��м��壬����CH3COOH�ڣ�C2H5O��3P=O�����¼�����H2O�õ�����ϩͪ������̼ԭ���ӻ����������sp2��sp��1mol��C2H5O��3P=O�����к��еĦҼ�����ĿΪ25NA��
��4����֪��̬NH3��H2O��HF��������ܺͽṹ��ͼ1��
| ���� | ���X-H��Y | ����kJ��mol-1 |
| ��HF��n | D-H��F | 28.1 |
| �� | O-H��O | 18.8 |
| ��NH3��n | N-H��N | 5.4 |

����H2O��HF��NH3�е����ν��͵�ԭ������ļ����ǣ�HF��n��������NH3��n����ƽ��ÿ�����Ӻ������������2������HF��n�ͣ�NH3��nֻ��1��������Ҫ�˷���������ܼ����DZ�����HF��n����NH3��n����
��5��̼����Ľṹ����ʯ���ƣ���Ӳ�Ƚ����ڽ��ʯ�����н�ǿ����ĥ���ܣ�̼���辧���ṹ��ÿ��̼ԭ����Χ�����������Ĺ�ԭ����4������̼ԭ�ӵȾ��������̼ԭ����12������֪̼���辧���߳�Ϊapm����ͼ2��1�Ź�ԭ�Ӻ�2��̼ԭ��֮��ľ���Ϊ$\frac{\sqrt{11}a}{4}$pm��̼������ܶ�Ϊ$\frac{1.6��1{0}^{32}}{{a}^{3}��{N}_{A}}$g/cm3��
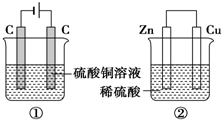 ��ͼ����װ�ã�����ʱת�Ƶĵ�������ȣ���Ӧ��ɺ������м���9.8gCu��OH��2���壬��Һǡ���ܻظ�ԭŨ�Ⱥ��������������Һ����ı仯������������ȷ���ǣ�������
��ͼ����װ�ã�����ʱת�Ƶĵ�������ȣ���Ӧ��ɺ������м���9.8gCu��OH��2���壬��Һǡ���ܻظ�ԭŨ�Ⱥ��������������Һ����ı仯������������ȷ���ǣ�������| A�� | ����������� ��=�� | |
| B�� | ���������������ӣ���������������С | |
| C�� | �缫��Ӧʽ������������Cu2++2e-�TCu����������2H++2e-�TH2�� | |
| D�� | ��Ӧ��ɺ���ҺPH���٣�7���ڣ�7 |
| A�� | ±�ص��ʵ��ܶ������� | |
| B�� | ���ǵ��⻯����ȶ�����˵���������Ӷ���ǿ | |
| C�� | ����ˮ�ӵ�KI������Һ�У���Һ���� | |
| D�� | ���ǵ�ԭ�Ӻ�����Ӳ�����˵���������Ӷ����� |
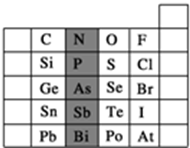 Ԫ�������ڱ��е�λ�ã���ӳ��Ԫ�ص�ԭ�ӽṹ��Ԫ�ص����ʣ���ͼ��Ԫ�����ڱ���һ���֣�
Ԫ�������ڱ��е�λ�ã���ӳ��Ԫ�ص�ԭ�ӽṹ��Ԫ�ص����ʣ���ͼ��Ԫ�����ڱ���һ���֣� ��
��
 ��
��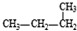
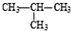
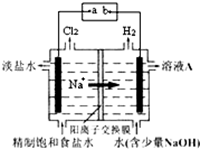 �ȼҵ�е�ⱥ��ʳ��ˮ��ԭ��ʾ��ͼ��ͼ��ʾ��
�ȼҵ�е�ⱥ��ʳ��ˮ��ԭ��ʾ��ͼ��ͼ��ʾ��