��Ŀ����
ij��ѧѧϰС��Ϊ�˽����ἰ���ε�ijЩ���ʺ���;������������ʵ��̽����

��ʵ��һ��̽��Ũ����������ԣ�
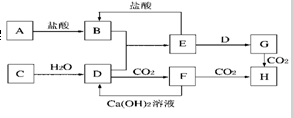
��1��д����ƿ�з�Ӧ�Ļ�ѧ����ʽ�� ��
��2����ͼ1��Bװ�ÿ�����̽��SO2�Ļ�ԭ�ԣ���B�����õ��Լ�Ϊ�� ����дһ�ּ��ɣ�����Bװ����Ʒ����Һ����B�е������� ��
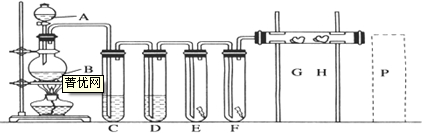
��ʵ��������飨NH4��2Fe��SO4��2?6H2O�еĽ������ӣ�
��3�����ϻ�ѧ����ͱ�Ҫ������˵����μ�������еĽ������ӣ��Լ���ԭ��������ȣ��� ��
��ʵ��������KHSO4��ȡH2O2����������������
�������ϵ�֪����ҵ��������ԭ����ȡ��ȡH2O2��
���ô˷���ȡһ��Ũ�ȵ�H2O2�����ⶨH2O2������������
��֪��2MnO4-+5H2O2+6H+=2Mn2++8H2O+5O2��
��ȡ10.00mL H2O2��Һ���ܶ�Ϊ1.00g/mL��������ƿ�м�ˮϡ�ͣ�����ϡ�����ữ��
����0.200 0mol/L KMnO4��Һ�ζ���
����ͬ�������ζ��ĴΣ�����KMnO4��Һ������ֱ�Ϊ��40.00mL��39.98mL��42.02mL��40.02mL��
��4���ø�ԭ����ȡH2O2�������缫��ӦʽΪ
��5�����в�����ʱ�������һ��KMnO4��Һ����Һ���Ϻ�ɫ��ʧ���������ŵζ��Ľ��У���Һ���Ϻ�ɫ��ʧ���ʼӿ죬�Ʋ���ԭ�������
��6����H2O2��Һ�����ʵ���������Ϊ ��

��ʵ��һ��̽��Ũ����������ԣ�
��1��д����ƿ�з�Ӧ�Ļ�ѧ����ʽ��
��2����ͼ1��Bװ�ÿ�����̽��SO2�Ļ�ԭ�ԣ���B�����õ��Լ�Ϊ��
��ʵ��������飨NH4��2Fe��SO4��2?6H2O�еĽ������ӣ�
��3�����ϻ�ѧ����ͱ�Ҫ������˵����μ�������еĽ������ӣ��Լ���ԭ��������ȣ���
��ʵ��������KHSO4��ȡH2O2����������������
�������ϵ�֪����ҵ��������ԭ����ȡ��ȡH2O2��
���ô˷���ȡһ��Ũ�ȵ�H2O2�����ⶨH2O2������������
��֪��2MnO4-+5H2O2+6H+=2Mn2++8H2O+5O2��
��ȡ10.00mL H2O2��Һ���ܶ�Ϊ1.00g/mL��������ƿ�м�ˮϡ�ͣ�����ϡ�����ữ��
����0.200 0mol/L KMnO4��Һ�ζ���
����ͬ�������ζ��ĴΣ�����KMnO4��Һ������ֱ�Ϊ��40.00mL��39.98mL��42.02mL��40.02mL��
��4���ø�ԭ����ȡH2O2�������缫��ӦʽΪ
��5�����в�����ʱ�������һ��KMnO4��Һ����Һ���Ϻ�ɫ��ʧ���������ŵζ��Ľ��У���Һ���Ϻ�ɫ��ʧ���ʼӿ죬�Ʋ���ԭ�������
��6����H2O2��Һ�����ʵ���������Ϊ
���㣺Ũ���������,̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ����
��������ʵ��һ����1��ͭ��Ũ�����ڼ��������·�Ӧ��������ͭ�����������ˮ��
��2��̽��SO2�Ļ�ԭ�ԣ���B�����õ��Լ�Ϊ���Ը��̼أ������������Ư���ԣ���ʹƷ����ɫ���������������������Ĵ��ڣ�
��ʵ���������Fe2+�ȼ�KSCN��Һ���ټ���ˮ��
��ʵ��������3�����ص������������ӷ���ʧ���ӵ�������Ӧ��
��4����˫��ˮ�ķֽ�����У����������������ã�
��5���������������ǿ�����ԣ����Խ�˫��ˮ������
��6�����ݷ�Ӧ��ԭ������ʽ���м��㣮
��2��̽��SO2�Ļ�ԭ�ԣ���B�����õ��Լ�Ϊ���Ը��̼أ������������Ư���ԣ���ʹƷ����ɫ���������������������Ĵ��ڣ�
��ʵ���������Fe2+�ȼ�KSCN��Һ���ټ���ˮ��
��ʵ��������3�����ص������������ӷ���ʧ���ӵ�������Ӧ��
��4����˫��ˮ�ķֽ�����У����������������ã�
��5���������������ǿ�����ԣ����Խ�˫��ˮ������
��6�����ݷ�Ӧ��ԭ������ʽ���м��㣮
���
�⣺��ʵ��һ����1��ͭ��Ũ�����ڼ��������·�Ӧ��������ͭ�����������ˮ����Cu+2H2SO4��Ũ��
CuSO4+SO2��+2H2O���ʴ�Ϊ��Cu+2H2SO4��Ũ��
CuSO4+SO2��+2H2O��
��2��̽��SO2�Ļ�ԭ�ԣ���B�����õ��Լ�Ϊ���Ը��̼أ������������Ư���ԣ���ʹƷ����ɫ���ʿ���Ʒ����Һ�������������Ĵ��ڣ��ʴ�Ϊ�����Ը�����أ�Ʒ����ɫ�ڣ�
��ʵ���������Fe2+�ȼ�KSCN��Һ���ټ���ˮ��������KSCN��Һ������˵��û��Fe3+���ټ���ˮ��Һ���ɫ˵����Fe2+����Ϊ������Fe2+������Fe3+��Fe3+����KSCN��Һ�Ժ�ɫ��
�ʴ�Ϊ��KSCN����ˮ��������Fe2+������Fe3+��Fe3+����KSCN��Һ�Ժ�ɫ��
��ʵ��������3����ⱥ��KHSO4��Һʱ�������������ӷ���ʧ���ӵ�������Ӧ���缫��ӦʽΪ2HSO4--2e-=S2O82-+2H+����2SO42--2e-=S2O82-�����ʴ�Ϊ��2HSO4--2e-=S2O82-+2H+����2SO42--2e-=S2O82-����
��4����˫��ˮ�ķֽ�����У�������ؾ��������ԣ���Ӧ�Ļ�ԭ�����������ӣ��������һ��KMnO4��Һ����Һ�Ϻ�ɫ��ʧ���������ŵζ�������Mn2+�����࣬��Һ�Ϻ�ɫ��ʧ���ʼӿ죬�ɼ������������������ã�
�ʴ�Ϊ�����������������ã�
��5���Ĵ�����KMnO4��Һ������ֱ�Ϊ40.00mL��39.98mL��42.02mL��40.02mL�����������Ϊ��Ч���ݣ�����������ƽ��ֵΪ��40.00mL�������ĸ������������0.2mol/L��0.04L=0.008mol����˫��ˮ�����ʵ���Ϊn����
2MnO4-+5H2O2+6H+=2Mn2++8H2O+5O2��
2 5
0.008mol n
���n=0.002mol������˫��ˮ������Ϊ��0.002mol��34g/mol=0.68g��˫��ˮ����������=
��100%=6.8%���ʴ�Ϊ��6.8%��
| ||
| ||
��2��̽��SO2�Ļ�ԭ�ԣ���B�����õ��Լ�Ϊ���Ը��̼أ������������Ư���ԣ���ʹƷ����ɫ���ʿ���Ʒ����Һ�������������Ĵ��ڣ��ʴ�Ϊ�����Ը�����أ�Ʒ����ɫ�ڣ�
��ʵ���������Fe2+�ȼ�KSCN��Һ���ټ���ˮ��������KSCN��Һ������˵��û��Fe3+���ټ���ˮ��Һ���ɫ˵����Fe2+����Ϊ������Fe2+������Fe3+��Fe3+����KSCN��Һ�Ժ�ɫ��
�ʴ�Ϊ��KSCN����ˮ��������Fe2+������Fe3+��Fe3+����KSCN��Һ�Ժ�ɫ��
��ʵ��������3����ⱥ��KHSO4��Һʱ�������������ӷ���ʧ���ӵ�������Ӧ���缫��ӦʽΪ2HSO4--2e-=S2O82-+2H+����2SO42--2e-=S2O82-�����ʴ�Ϊ��2HSO4--2e-=S2O82-+2H+����2SO42--2e-=S2O82-����
��4����˫��ˮ�ķֽ�����У�������ؾ��������ԣ���Ӧ�Ļ�ԭ�����������ӣ��������һ��KMnO4��Һ����Һ�Ϻ�ɫ��ʧ���������ŵζ�������Mn2+�����࣬��Һ�Ϻ�ɫ��ʧ���ʼӿ죬�ɼ������������������ã�
�ʴ�Ϊ�����������������ã�
��5���Ĵ�����KMnO4��Һ������ֱ�Ϊ40.00mL��39.98mL��42.02mL��40.02mL�����������Ϊ��Ч���ݣ�����������ƽ��ֵΪ��40.00mL�������ĸ������������0.2mol/L��0.04L=0.008mol����˫��ˮ�����ʵ���Ϊn����
2MnO4-+5H2O2+6H+=2Mn2++8H2O+5O2��
2 5
0.008mol n
���n=0.002mol������˫��ˮ������Ϊ��0.002mol��34g/mol=0.68g��˫��ˮ����������=
| 0.68g |
| 10.00mL��1.00g/mL |
���������⿼����Ũ������ͭ�ķ�Ӧ���к͵ζ��ļ��㣬��Ŀ�ϻ������к͵ζ�����ʱ��Ҫȡ����ʵ���ƽ��ֵ�����в���������Ҫ��ȥ��

��ϰ��ϵ�д�
�����Ŀ
��һС�������Ͷ�뵽CuSO4��Һ�У������ܵõ��IJ����ǣ�������
| A��Cu |
| B��H2 |
| C��Cu��OH��2 |
| D��Na2SO4 |
������������ָ����Һ���ܴ���������ǣ�������
| A������������Һ�У�Cu2+��K+��SO42-��S2- |
| B�����뱽������ɫ����Һ��K+��NH4+��Cl-��SCN- |
| C��1.0 mol?L-1��KNO3��Һ�У�Na+��Fe2+��Cl-��SO42- |
| D�����ܽ�Al2O3����Һ�У�Mg2+��Fe3+��AlO2-��HCO3- |
������Һ�������ɺ����գ����������ʹ�����ǣ�������
| A��AlCl3 |
| B��KHCO3 |
| C��Fe2��SO4��3 |
| D��NH4HCO3 |

 �ش��������⣺
�ش��������⣺