��Ŀ����
��1 L�ܱ������з�����Ӧ��4NH3(g)+5O2(g) 4NO(g)+6H2O(g)
4NO(g)+6H2O(g)
��H=��Q kJ��mol-1��Q>O���������ڲ������ʵ����ʵ���Ũ�����±���
| ʱ��/Ũ�� | c(NH3) (mol/L) | c(O2 ) (mol/L) | c(NO) (mol/L) |
| ��ʼ | 0.8 | 1.6 | 0 |
| ��2min | 0.6 | a | 0.2 |
| ��4min | 0.3 | 0.975 | 0.5 |
| ��6min | 0.3 | 0.975 | 0.5 |
| ��8min | 0.7 | 1.475 | 0.1 |
| ��10min | 0.7 | 1.475 | 0.1 |
A����Ӧ�ڵ�2 min����4minʱ��O2��ƽ����Ӧ����Ϊ0.1875 mol/(L��min)
B����Ӧ�ڵ�2 minʱ�ı���ijһ������������������ʹ�ô����������¶�
C����4 minʱ����8 minʱ�ֱ�ﵽ��ѧƽ�⣬��ƽ�ⳣ������ͬ
D���ڿ�ʼ��Ӧ��ǰ2 min�ڣ��÷�Ӧ�ġ�H=��0.05Q kJ��mol-1
BD
����

 ��Уͨ��֤��Ч��ҵϵ�д�
��Уͨ��֤��Ч��ҵϵ�д���˫ѡ����1 L�ܱ������з�����Ӧ��4NH3(g)+5O2(g) 4NO(g)+6H2O(g) ��H=��Q kJ��mol��1��Q>O���������ڲ������ʵ����ʵ���Ũ�����±���
4NO(g)+6H2O(g) ��H=��Q kJ��mol��1��Q>O���������ڲ������ʵ����ʵ���Ũ�����±���
|
ʱ�� |
c(NH3) (mol/L) |
c(O2 ) (mol/L) |
c(NO) (mol/L) |
|
��ʼ |
0.8 |
1.6 |
0 |
|
��2min |
0.6 |
a |
0.2 |
|
��4min |
0.3 |
0.975 |
0.5 |
|
��6min |
0.3 |
0.975 |
0.5 |
|
��8min |
0.7 |
1.475 |
0.1 |
|
��10min |
0.7 |
1.475 |
0.1 |
����˵���������
A����Ӧ�ڵ�2 min����4minʱ��O2 ��ƽ����Ӧ����Ϊ0.1875 mol/(L��min)
B����Ӧ�ڵ�2 minʱ�ı���ijһ������������������ʹ�ô����������¶�
C����4 minʱ����8 minʱ�ֱ�ﵽ��ѧƽ�⣬��ƽ�ⳣ������ͬ
D���ڿ�ʼ��Ӧ��ǰ2 min�ڣ��÷�Ӧ�ġ�H=��0.05Q kJ��mol��1
��1 L�ܱ������з�����Ӧ��4NH3(g)+5O2(g)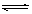 4NO(g)+6H2O(g)
4NO(g)+6H2O(g)
��H=��Q kJ��mol-1��Q>O���������ڲ������ʵ����ʵ���Ũ�����±���
|
ʱ��/Ũ�� |
c(NH3) (mol/L) |
c(O2 ) (mol/L) |
c(NO) (mol/L) |
|
��ʼ |
0.8 |
1.6 |
0 |
|
��2min |
0.6 |
a |
0.2 |
|
��4min |
0.3 |
0.975 |
0.5 |
|
��6min |
0.3 |
0.975 |
0.5 |
|
��8min |
0.7 |
1.475 |
0.1 |
|
��10min |
0.7 |
1.475 |
0.1 |
����˵���������
A����Ӧ�ڵ�2 min����4minʱ��O2 ��ƽ����Ӧ����Ϊ0.1875 mol/(L��min)
B����Ӧ�ڵ�2 minʱ�ı���ijһ������������������ʹ�ô����������¶�
C����4 minʱ����8 minʱ�ֱ�ﵽ��ѧƽ�⣬��ƽ�ⳣ������ͬ
D���ڿ�ʼ��Ӧ��ǰ2 min�ڣ��÷�Ӧ�ġ�H=��0.05Q kJ��mol-1
 2C(g)����ʼʱ����4 mol A��6 mol B����2 minĩʱ���C�����ʵ�����2 mol��
2C(g)����ʼʱ����4 mol A��6 mol B����2 minĩʱ���C�����ʵ�����2 mol�� Ũ��
Ũ�� 2C(g)����ʼʱ����4 mol A��6 mol B����2 minĩʱ���C�����ʵ�����2 mol��
2C(g)����ʼʱ����4 mol A��6 mol B����2 minĩʱ���C�����ʵ�����2 mol��