��Ŀ����
12����ѧ��Ӧԭ������ѧ��ѧѧϰ����Ҫ���ݣ���ش��������⣺
��1�������ж���ȷ���Ǣ٢ۣ�
��CH4��g��+2O2��g���TCO2��g��+2H2O��l����H1
CH4��g��+2O2��g���TCO2��g��+2H2O��g����H2���H1����H2
��H2��g��+$\frac{1}{2}$O2��g���TH2O��l����H1
2H2��g��+O2��g���T2H2O��l����H2���H1����H2
��t��ʱ����һ�������£���1mol SO2��1mol O2�ֱ����ں��ݺͺ�ѹ�������ܱ������У��ﵽƽ��״̬ʱ��Ӧ�ų��������ֱ�ΪQ1��Q2��Q1��Q2
��CaCO3��s���TCaO��s��+CO2��g����H1
CaO��s��+H2O��l���TCa��OH��2��s����H2���H1����H2��
��2������������ԭ��ӦZn��s��+Cu2+��aq���TZn2+��aq��+Cu��s��
��Ƶ�ԭ�����ͼ1��ʾ��
��ش��������⣺
��ͭ�缫�����ĵ缫��ӦΪCu2++2e-�TCu����Һ��Cu2+�������ƶ���
��3����һ���¡������ܱ������г���1 mol CO2��3 mol H2��һ�������·�����Ӧ��CO2��g��+3H2��g��?CH3OH ��g��+H2O��g����H=-49.0 kJ/mol
ij�ַ�Ӧ����������Ũ����ʱ��仯��ͼ2��ʾ���ش��������⣺
��Y�Ļ�ѧʽ��CO2
�ڷ�Ӧ���е�3minʱ��v����v�����������������=��������Ӧǰ3min��H2��ƽ����Ӧ����v��H2��=0.5mol•L-1•min-1��
����֤���÷�Ӧ�ﵽ��ѧƽ��״̬��������ACD
A�������ڸ����������������ֲ���
B����������ܶȲ���
C��3v����CH3OH��=v����H2��
D����������ƽ����Է�����������
E��CO2��ת����Ϊ70%
F�����������CO2��H2�������Ϊ1�s3
�������¶��£���ӦCH3OH ��g��+H2O��g��?CO2��g��+3H2��g����ƽ�ⳣ��K=0.19������������2λС������
��������Ӧ�ﵽƽ�����������ͬʱ����0.1mol CO2��0.3mol H2O ��g������ʱƽ�⽫���ң�����������ҡ��������ƶ���
��4������ʱ����20 mL 0.1 mol/L�Ĵ�����Һ�в��ϵ���0.1 mol/L��NaOH��Һ����Һ��pH�仯������NaOH��Һ����Ĺ�ϵ��ͼ3��ʾ�������й���Һ�����ӡ�����Ũ�ȴ�С��ϵ����������ȷ���Ǣڢۣ�
��a��ʱ��c��CH3COOH����c��CH3COO-���� c��Na+���� c��H+����c��OH-��
��b��ʱ��c��Na+��=c��CH3COO-����c��H+��=c��OH-��
��c��ʱ��c��OH-��=c��CH3COOH��+c��H+��
��d��ʱ��c��Na+���� c��CH3COO-���� c��OH-����c��H+��
���� ��1���������ʵ�״̬Ӱ�췴Ӧ�ȵ���ֵ����Ӧ�ȵķ��������ȡ����ȵĹ�ϵ����Ӧ�ȵ���ֵ�뻯ѧ����ʽǰ���ϵ�����������ش�ע��ˮ������ΪҺ̬�Ƿ��ȵģ����ʵķֽⷴӦ�����ȵģ����ʵĻ��Ϸ�Ӧ������Ƿ��ȵģ����ȷ�Ӧ���ʱ�Ϊ��ֵ�Ƚϴ�С��
��2��װ��ͼ������֪Ϊͭпԭ��أ�ͭ��������п��ԭ��صĸ������������Һ��ͭ�����������õ���������ͭ��������пʧ���ӷ���������Ӧ��ԭ���������������������������������
��3������ͼ��֪��Ӧ���е�3minʱ�����ĵ�Y�����ɵ�X��Ϊ0.5mol��˵�����ߵļ�������ͬ�����ݷ���ʽ�жϣ�
�ڷ�Ӧ���е�3minʱ����Ӧδ����ƽ��״̬�����ݶ�����̼��Ũ�ȱ仯���������Ũ�ȱ仯������Ӧ���ʣ�
����һ�������£�����ѧ��Ӧ�ﵽƽ��״̬ʱ�����淴Ӧ������ȣ������ʵ�Ũ�Ȳ��䣬�ɴ�������һЩ������Ҳ���䣻
���ȼ����ƽ�ⳣ��K����ͼ��֪���ﵽƽ��״̬ʱ������̼ת����0.75mol/L����H2ת����0.75mol/L��3=2.25mol/L�����ɵ�c��CH3OH��=c��H2O��=0.75mol/L����ƽ��ʱc��CO2��=0.25mol/L��c��H2��=0.75mol/L��c��CH3OH��=0.75mol/L��c��H2O��=0.75mol/L���ݴ˼��㻯ѧ��ӦCO2��g��+3H2��g��?CH3OH ��g��+H2O��g����ƽ�ⳣ�������淴Ӧ��ƽ�ⳣ����Ϊ������ϵ��
����������ͬʱ����1molCO2��1mol H2O��g�����ٸ���Qc��K�Ĵ�С��ϵ�жϷ�Ӧ���еķ���
��4���٣�a����ҺΪCH3COOH��CH3COONa�Ļ�����Һ�����ԣ�c��H+����c��OH-����˵������ĵ���̶ȴ��ڴ�������ӵ�ˮ��̶ȣ�
�ڣ�b��ʱ��Һ��pH=7��c��H+��=c��OH-�������ݵ���غ��֪c��Na+��=c��CH3COO-����
�ۣ�c��ʱ��ǡ�÷�Ӧ���ɴ�������Һ�����ݵ���غ�������غ�����жϣ�
�ܣ�d���������ƹ�����ΪNaOH��CH3COONa�Ļ�����Һ�ʼ��ԣ�
��� ��1������CH4��g��+2O2��g��=CO2��g��+2H2O��g����ȼ�շ�Ӧ����Ӧ�����ȣ�������0��
CH4��g��+2O2��g��=CO2��g��+2H2O��l����H1��CH4��g��+2O2��g��=CO2��g��+2H2O��g����H2����ˮ����̬���Һ̬���ų����������ԡ�H1����H2���ʢ���ȷ��
��H2��g��+$\frac{1}{2}$O2��g���TH2O��l����H1��2H2��g��+O2��g���T2H2O��l����H2��������ȼ���Ƿ��ȵģ������ʱ��Ǹ�ֵ��ϵ���ӱ����ʱ���ֵҲ�ӱ������ԡ�H2=2��H1��0�����ԡ�H2����H1���ʢڴ���
�ۡ�ʱ����һ�������£���1 mol SO2��1 mol O2�ֱ����ں��ݺͺ�ѹ�������ܱ������У���ѹ�ܱ������൱���ں��ݵĻ����������ƶ��ˣ����Ժ�P�£��ﵽƽ��״̬ʱ�ų����������࣬�ﵽƽ��״̬ʱ�ų��������ֱ�ΪQ1��Q2��Q1��Q2���ʢ���ȷ��
��CaCO3��s���TCaO��s��+CO2��g����H1��0��CaO��s��+H2O��l���TCa��OH��2��s����H2��0�����ԡ�H1����H2���ʢܴ���
�ʴ�Ϊ���٢ۣ�
��2��װ��ͼ������֪Ϊͭпԭ��أ�ͭ��������п��ԭ��صĸ������������Һ��ͭ�����������õ���������ͭ��������пʧ���ӷ���������Ӧ��ԭ���������������������������������
��ͭ�缫�����ĵ缫��ӦΪ��Cu2++2e-�TCu���ʴ�Ϊ��Cu2++2e-�TCu��
������������֪ԭ���������������������������������
�ʴ�Ϊ������
��3������ͼ��֪��Ӧ���е�3minʱ�����ĵ�Y�����ɵ�X��Ϊ0.5mol��˵�����ߵļ�������ͬ����֪CO2��g��+3H2��g��?CH3OH��g��+H2O��g����
��Ӧ��YΪCO2���ʴ�Ϊ��CO2��
�ڷ�Ӧ���е�3minʱ����Ӧδ����ƽ��״̬����Ӧ��������У�����v����v������Ӧǰ3min�����ĵĶ�����̼Ϊ1.00-0.50=0.50mol/L�������ĵ�����Ϊ
0.50mol/L��3=1.50mol/L����v��H2��=$\frac{1.50mol/L}{3min}$=0.5mol•L-1•min-1��
�ʴ�Ϊ������0.5��
����һ�������£�����ѧ��Ӧ�ﵽƽ��״̬ʱ�����淴Ӧ������ȣ������ʵ�Ũ�Ȳ��䣬
A�������ڸ����������ȱ��ֲ��䣬˵�������ʵ������ֲ��䣬����֤���Ѿ��ﵽƽ��״̬����A��ȷ��
B����������������䣬�����غ㣬���Ի��������ܶ�ʼ�ղ��䣬���Բ���֤���Ѿ��ﵽƽ��״̬����B����
C���ﵽƽ��״̬ʱ����ͬ���ʵķ�Ӧ����֮�ȵ��ڼ�����֮�ȣ�����ƽ��״̬������3v����CH3OH��=v����H2�� ʱ��˵���Ѿ��ﵽƽ��״̬����C��ȷ��
D����ͼ��֪���ﵽƽ��״̬ʱ������̼ת����0.75mol/L����H2ת����0.75mol/L��3=2.25mol/L������������ת����Ϊ $\frac{2.25}{3}$��100%=75%����D��ȷ��
�ʴ�Ϊ��ACD��
����ͼ��֪���ﵽƽ��״̬ʱ������̼ת����0.75mol/L����H2ת����0.75mol/L��3=2.25mol/L�����ɵ�c��CH3OH��=c��H2O��=0.75mol/L����ƽ��ʱc��CO2��=0.25mol/L��c��H2��=0.75mol/L��c��CH3OH��=0.75mol/L��c��H2O��=0.75mol/L��
��K=$\frac{[C{H}_{3}COOH][{H}_{2}O]}{[C{O}_{2}][{H}_{2}]^{3}}$=$\frac{0.75��0.75}{0.25��0.7{5}^{3}}$=5.33����ӦCH3OH��g��+H2O��g��?CO2��g��+3H2��g���Ƿ�ӦCO2��g��+3H2��g��?CH3OH��g��+H2O��g�����淴Ӧ�����ߵ�ƽ�ⳣ����Ϊ�����Ĺ�ϵ����K��=$\frac{1}{K}$=$\frac{1}{5.33}$=0.19���ʴ�Ϊ��0.19��
����������ͬʱ����1molCO2��1mol H2O��g������Qc=$\frac{0.75����1+0.75��}{��0.25+1����0.7{5}^{3}}$=2.49��K=5.33����Ӧδ����ƽ��״̬�����Է�Ӧ���ҽ��У�
�ʴ�Ϊ�����ң�
��4���٣�a��ʱ�����������ҺΪCH3COOH��CH3COONa�Ļ�����Һ�����ԣ���������ӵ�ˮ��̶�С�ڴ���ĵ���̶ȣ���c��CH3COO-����c��CH3COOH�����ʢٴ���
�ڣ�b��ʱ������Һ����غ��֪��Һ��Ӧ����c��Na+��+c��H+��=c��CH3COO-��+c��OH-������Һ�����ԣ�Ӧ��c��H+��=c��OH-������c��Na+��=c��CH3COO-����c��Na+��=c��CH3COO-����c��H+��=c��OH-�����ʢ���ȷ��
�ۣ�c��ʱ���������������ǡ�÷�Ӧ���ɴ�������Һ����������Һ�У����ݵ���غ�ɵã���c��Na+��+c��H+��=c��CH3COO-��+c��OH-�������������غ�ɵã���c��Na+��=c��CH3COO-��+c��CH3COOH�������ڴ��ˢٿɵã�c��OH-��=c��CH3COOH��+c��H+����Ϊ�����غ㣬�ʢ���ȷ��
�ܣ�d��ΪNaOH��CH3COONa�Ļ�����Һ�ʼ��ԣ�����CH3COO-��������ˮ�⣬����c��Na+����c��OH-����c��CH3COO-����c��H+�����ʢܴ���
�ʴ�Ϊ���ڢۣ�
���� ���⿼�������ʷ�Ӧ�����仯�����жϣ���Ӧ���ʼ��㡢��ѧƽ��ļ�����Ӱ�����ء�ƽ�ⳣ���ļ��㡢��Ӧ������жϣ�����ϵ��жϺ�����Ũ�ȴ�С�Ƚϵȣ������ڿ���ѧ�������ͽ���������������Ŀ�ѶȽϴ�ע��������Һ���������ҺpH�Ĺ�ϵ���ܹ����ݵ���غ㡢�����غ㡢�ε�ˮ���ж���Һ������Ũ�ȴ�С��

 ����ν����Ž̲��㽭���̴�ѧ������ϵ�д�
����ν����Ž̲��㽭���̴�ѧ������ϵ�д� �����Ļ������������������ϵ�д�
�����Ļ������������������ϵ�д�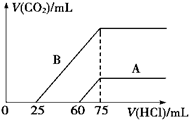 ��1��ȡ�����ʵ���Ũ�ȵ�NaOH��Һ����A��B��ÿ��10mL���ֱ���A��B��ͨ�벻������CO2���ټ���������Һ����μ���0.1mol/L�����ᣬ��״���²�����CO2������������������֮��Ĺ�ϵ��ͼ��ʾ���Իش��������⣺
��1��ȡ�����ʵ���Ũ�ȵ�NaOH��Һ����A��B��ÿ��10mL���ֱ���A��B��ͨ�벻������CO2���ټ���������Һ����μ���0.1mol/L�����ᣬ��״���²�����CO2������������������֮��Ĺ�ϵ��ͼ��ʾ���Իش��������⣺��ԭNaOH��Һ�����ʵ���Ũ��Ϊ0.75mol/L��
������A������ԭNaOH��Һ��ͨ��CO2��������Һ�е����ʳɷ���Na2CO3��NaOH�������ʵ���֮��Ϊ1��3��
������B������ԭNaOH��Һ��ͨ��CO2��������Һ����������CO2�����������״���������ֵΪ112mL��
��2�����мס�����ƿ��ɫ��Һ����֪���ǿ���ΪAlCl3��Һ��NaOH��Һ���ֱַ�һ������ļס�������Һ��ϣ�����������±���ʾ����ش�
| ʵ��� | ʵ��� | ʵ��� | |
| ȡ��ƿ��Һ���� | 400mL | 120mL | 120mL |
| ȡ��ƿ��Һ���� | 120mL | 440mL | 400mL |
| ���ɳ������� | 1.56g | 1.56g | 3.12g |
����ƿ��Һ�����ʵ���Ũ��Ϊ0.5mol/L��
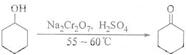
������������ͪ��ˮ�IJ����������ʼ��±���
| ���� | �е㣨�棩 | �ܶȣ�g-cm-3��20�棩 | �ܽ��� |
| ������ | 161.1��97.8��* | 0.9624 | ������ˮ |
| ����ͪ | 155.6��95��* | 0.9478 | ����ˮ |
| ˮ | 100.0 | 0.9982 |
��1������Na2Cr2O7��Һ������������Ӧ�ġ�H��0����Ӧ���ҽ�������ϵ�¶�Ѹ������������Ӧ���࣮ʵ���н�����Na2Cr2O7��Һ�ӵ�ʢ�л���������ƿ�У���55-60����з�Ӧ����Ӧ��ɺ�������ˮ�������ռ�95-100�����֣��õ���Ҫ�ĺ�����ͪ��ˮ�Ļ���
������Na2Cr2O7��Һ�ļ��Ϸ�ʽΪ�����μӣ�
�������ܷ��뻷��ͪ��ˮ��ԭ���ǻ���ͪ��ˮ�γɾ��й̶���ɵĻ����һ��������
��2������ͪ���ᴿ��Ҫ��������һϵ�еIJ�����
a�����ռ�151-156�����֣�b ���ˣ�c ���ռ���������м�NaCl���������ͣ����ã���Һ��d ������ˮMgSO4���壬��ȥ�л���������ˮ��
��������������ȷ˳����cdba������ĸ����
����������b��c��ʹ�õIJ����������ձ�����ƿ���������⣬����©������Һ©����
������������c�У�����NaCl���������������ˮ����ܶȣ������ڷֲ㣮
��3�����ú������ͼ����ȷ������ͪ���Ӻ������������ʻ���
| A�� | ������Һ�е����ˮ--����ɫ | B�� | �廯������ֽ�--���ɫ | ||
| C�� | ������Һ�е���Fe 3+--����ɫ | D�� | ���������о���--���ɫ |
̽��I ���CN-Ũ��
���ϣ����������·������ӷ�Ӧ��2CN-+5H202+90H-�T2CO32-+N2+6H20
ʵ��װ����ͼ1�����м��ȡ��г�װ��ʡ�ԣ�������������ˮ���������ӷ�Ӧ��
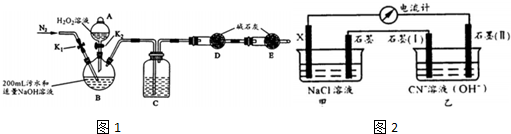
��1������ҩƷ֮ǰ�IJ����Ǽ��װ�������ԣ�C���Լ���ŨH2S04
��2��ʵ�鲽�����£�
| ����1 | �ر�K1����K2����������H2O2��Һ����B���ȣ���ַ�Ӧ��ֹͣ���� |
| ����2 | �ر�K2����ע��������Bװ�õĽ���ע�� ϡH2S04��Һ |
| ����3 | ��K1��ͨ��N2 |
��4��Ϊ��ʹ������ȷ��������2���в���Ҫ�ر�ע��һЩ�����д������һ����ϡH2S04ʱҪ����ע�루Ҫ����ͨ��N2���B����ʱ�¶Ȳ��ܹ������ϡH2SO4��Ҫ��������
̽����绯ѧ������CN-��Ӱ����������ͼ2װ��ģ��绯ѧ��ʵ�飬�йؽ�������
| ʵ����� | �缫��X�� | NaCl��ҺŨ�� ��mol/L�� | ����ʯī����ͨ������ | �����ƶ��� ��A�� |
| ��1�� | Fe | 0.1 | ���� | I |
| ��2�� | Al | 0.1 | ���� | 1.5I |
| ��3�� | Fe | 0.1 | O2 | 2I |
| ��4�� | Al | 0.5 | ���� | 1.6I |
��X�缫���ƣ�������
��ʵ�飨1����X����Zn��������ƶ����ķ�ΧΪI��1.5I
������װ�������������������ʣ���缫����ʽΪ2CN-+12OH--10e-=2CO32-+N2+6H2O����ʵ��˵���绯ѧ������CN-��Ӱ�촦�����ʵ������и����Ľ������Ϻͼ��е���ʵ�Ũ�ȣ�������������
 ��
��
 ��
��