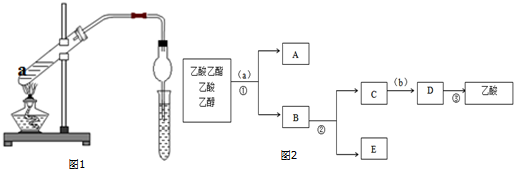
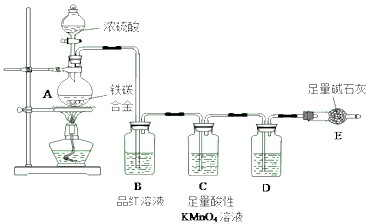
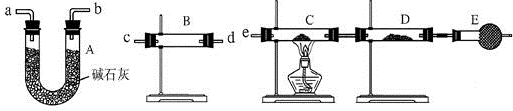
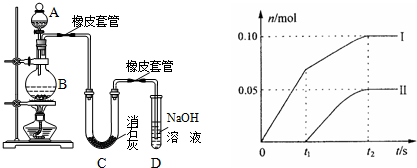
��Ŀ����
ij�Ȼ�����Ʒ��������FeCl2���ʣ���Ҫ�ⶨ������Ԫ�ص�����������ʵ�鰴���²�����У�
��Ʒa g250.00mL��Һ25.0mL��Һ��������ɫ����b g����ˮ������ˮ�ڹ�����ˮ����ϴ������
������������̣��ش��������⣺
��1������I��������Һ�����õ��IJ����������ձ����������⣬�������� �� �������������ƣ�
��2�����в�������ʹ������ҺŨ��ƫС���� ����д��ţ���
��δϴ���ձ��Ͳ�����
�ڶ���ʱ��������ƿ�Ŀ̶���
������Һǰ����ƿ������������ˮ
��ҡ�Ⱥ���Һ����ڿ̶��ߺ������ˮ����Һ����̶�������
��3����д��������ˮ���������ӷ���ʽ ��
��4����������Ƿ��Ѿ�ϴ�Ӹɾ��IJ����� ��
��5����ԭ��Ʒ����aΪ50g�����Ⱥ����ɫ��������bΪ3g������Ʒ����Ԫ�ص����������� ��

��Ʒa g250.00mL��Һ25.0mL��Һ��������ɫ����b g����ˮ������ˮ�ڹ�����ˮ����ϴ������
������������̣��ش��������⣺
��1������I��������Һ�����õ��IJ����������ձ����������⣬��������
��2�����в�������ʹ������ҺŨ��ƫС����
��δϴ���ձ��Ͳ�����
�ڶ���ʱ��������ƿ�Ŀ̶���
������Һǰ����ƿ������������ˮ
��ҡ�Ⱥ���Һ����ڿ̶��ߺ������ˮ����Һ����̶�������
��3����д��������ˮ���������ӷ���ʽ
��4����������Ƿ��Ѿ�ϴ�Ӹɾ��IJ�����
��5����ԭ��Ʒ����aΪ50g�����Ⱥ����ɫ��������bΪ3g������Ʒ����Ԫ�ص�����������
���㣺̽�����ʵ���ɻ�������ʵĺ���,�������������������,����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺������Ҫ�Ľ������仯����
��������ʵ��Ŀ���Dzⶨ����������������ȡ�ķ�����ʹ��Ʒ�ܽ⡢��Ӧ������������������Ȼ��ͨ������������������������������
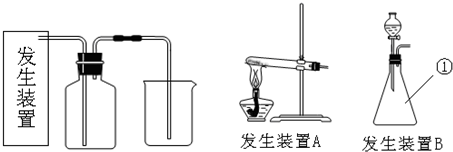
��1����ͼ��֪������I�ǽ��������ᷴӦ����Һϡ�ͳ�250.00mL��Һ������Ҫ250mL������ƿ��������Ҫ��ͷ�ιܣ�
��2���������������ʵ����ʵ��������Һ�������Ӱ�죬����c=
�����жϣ�
��3������ˮ���ǰ�+2��������Ϊ+3������
��4����Һ�д����Ȼ�泥����������ữ����������Һ�������һ��ϴ��Һ���Ƿ���������ӣ����жϳ����Ƿ�ϴ����
��5������Ԫ�������غ㣬������ɫ�����е���������Ʒ���������ݻ�ѧʽ������Ԫ�ص����������������������Ķ������ԭ��������Ʒ����Ԫ�ص�����������
��1����ͼ��֪������I�ǽ��������ᷴӦ����Һϡ�ͳ�250.00mL��Һ������Ҫ250mL������ƿ��������Ҫ��ͷ�ιܣ�
��2���������������ʵ����ʵ��������Һ�������Ӱ�죬����c=
| n |
| V |
��3������ˮ���ǰ�+2��������Ϊ+3������
��4����Һ�д����Ȼ�泥����������ữ����������Һ�������һ��ϴ��Һ���Ƿ���������ӣ����жϳ����Ƿ�ϴ����
��5������Ԫ�������غ㣬������ɫ�����е���������Ʒ���������ݻ�ѧʽ������Ԫ�ص����������������������Ķ������ԭ��������Ʒ����Ԫ�ص�����������
���
�⣺��1����ͼ��֪������I�ǽ��������ᷴӦ����Һϡ�ͳ�250.00mL��Һ����Ҫ250mL����ƿ������Ҫ�ý�ͷ�ιܶ��ݣ��ʴ�Ϊ��250mL����ƿ����ͷ�ιܣ�
��2����δϴ���ձ��Ͳ�������ת�Ƶ�����ƿ�����ʵ�������С��������ҺŨ��ƫ�ͣ��ʢ���ȷ��
�ڶ���ʱ��������ƿ�Ŀ̶��ߣ�ʹ��Һ�����ƫ�ͣ�������ҺŨ��ƫ�ߣ��ʢڴ���
�۶���ʱ��Ҫ��������ˮ������Һǰ����ƿ������������ˮ��Ӱ�죬�ʢ۴���
�ܼ�����ˮ����Һ����̶������У�ʹ��Һ���������������ҺŨ��ƫ�ͣ��ʢ���ȷ��
�ʴ�Ϊ���٢ܣ�
��3������ˮ������+2������Ϊ+3�ۣ�������ӦΪ��2Fe 2++Cl2=2Fe 3++2Cl-��
�ʴ�Ϊ��2Fe 2++Cl2=2Fe 3++2Cl-��
��4����Һ�д����Ȼ�泥����������ữ����������Һ�������һ��ϴ��Һ���Ƿ���������ӣ�
�ʴ�Ϊ��ȡ���һ��ϴ��Һ���Թ��У��μ�������������Һ�����ް�ɫ�������ɣ���֤����ϴ�Ӹɾ���
��5������Ԫ�������غ㣬������ɫ�����е���������Ʒ������Fe2O3������Ϊ3g�����ڲμӷ�Ӧ����Һֻȡ������Һ��
�������Ԫ�ص�����Ϊ30g��
=21g����Ʒ����Ԫ�ص�����������
��100%=42%���ʴ�Ϊ��42%��
��2����δϴ���ձ��Ͳ�������ת�Ƶ�����ƿ�����ʵ�������С��������ҺŨ��ƫ�ͣ��ʢ���ȷ��
�ڶ���ʱ��������ƿ�Ŀ̶��ߣ�ʹ��Һ�����ƫ�ͣ�������ҺŨ��ƫ�ߣ��ʢڴ���
�۶���ʱ��Ҫ��������ˮ������Һǰ����ƿ������������ˮ��Ӱ�죬�ʢ۴���
�ܼ�����ˮ����Һ����̶������У�ʹ��Һ���������������ҺŨ��ƫ�ͣ��ʢ���ȷ��
�ʴ�Ϊ���٢ܣ�
��3������ˮ������+2������Ϊ+3�ۣ�������ӦΪ��2Fe 2++Cl2=2Fe 3++2Cl-��
�ʴ�Ϊ��2Fe 2++Cl2=2Fe 3++2Cl-��
��4����Һ�д����Ȼ�泥����������ữ����������Һ�������һ��ϴ��Һ���Ƿ���������ӣ�
�ʴ�Ϊ��ȡ���һ��ϴ��Һ���Թ��У��μ�������������Һ�����ް�ɫ�������ɣ���֤����ϴ�Ӹɾ���
��5������Ԫ�������غ㣬������ɫ�����е���������Ʒ������Fe2O3������Ϊ3g�����ڲμӷ�Ӧ����Һֻȡ������Һ��
| 1 |
| 10 |
| 112 |
| 160 |
| 21 |
| 50 |
���������⿼����Һ���ơ����Ӽ��顢��ʵ�����������������������ѧ����ȣ��Ѷ��еȣ�����ⶨԭ���ǽ���Ĺؼ����Ƕ���ѧ֪ʶ���ۺ����ã���Ҫѧ��������ʵ�Ļ���������֪ʶ������������������

��ϰ��ϵ�д�
�����Ŀ
ʵ������һƿ���õİ�ɫK2SO3��ĩ��Ϊȷ�����Ƿ���������ɷ֣�Ԫ�ط���������ĩ��K��SԪ�ص�������Ϊ39��16�����н�����ȷ���ǣ�������
| A�����ݲ�õ�Ԫ�������ȿ��Ʋ�÷�ĩΪ������ |
| B������ĩ����ˮ�������Ȼ������а�ɫ�������ɣ�֤��ԭ��ĩ��K2SO4 |
| C������ĩ���������У��������ݣ�֤��K2SO3δ���� |
| D������ĩ����ˮ�������Ȼ������������ᣬ�а�ɫ�������������ɣ�֤��ԭ��ĩ��K2SO4��K2SO3�Ļ���� |
���и��������ܴ���������ǣ�������
| A��Cu2+��NO3-��Cl-��K+ |
| B��Fe3+��SO4-��SCN-��Na+ |
| C��Al3+��K+��AlO2-��Cl- |
| D��Fe2+��NO3-��Cl-��H+ |