��Ŀ����
11��2015��10���й�ҩѧ�����������������أ�һ����������ű����ҩ������ŵ��������ҽѧ���������أ�C15H22O5���Ľṹ��ͼ1��ʾ����ش��������⣺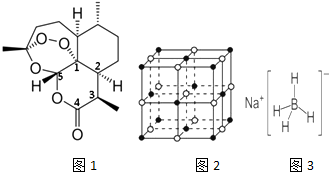
��1����������ص�����Ԫ�ص縺���ɴ�С������O��C��H���ڻ�̬Oԭ���У��������3�������෴�ĵ��ӣ�
��2�����й��������ص�˵����ȷ����a������ţ���
a���������мȴ��ڼ��Լ��ִ��ڷǼ��Լ�
b���������ط����У�����̼ԭ�Ӿ�����ͬһƽ��
c��ͼ�����ֱ�ʶ�����̼ԭ�Ӿ�ֻ�ԦҼ�������ԭ�ӳɼ�
��3����ȷ�������ؽṹ�Ĺ����У��ɲ���NaBH4��Ϊ��ԭ�������Ʊ�����Ϊ��4NaH+B��OCH3��3��NaBH4+3CH3ONa
��NaHΪ���Ӿ��壬��ͼ2��NaH�����ṹ����NaH�������λ����6���������ⳤΪa��Naԭ�Ӽ���С�˼��Ϊ$\frac{\sqrt{2}}{2}$a��
��B��OCH3��3��B���õ��ӻ�������sp2��д��������B��OCH3��3������ͬ�ռ乹�͵ķ��ӻ�����SO3��CO32-��
��NaBH4�ṹ��ͼ3��ʾ���ṹ�д��ڵ������������Ӽ�����λ�������ۼ���
���� ��1����������ص�����Ԫ��ΪH��C��O�����ݵ縺�Եĵݱ�����жϵ縺�ԵĴ�С˳���ڻ�̬Oԭ���У�������8�ĵ��ӣ�������3������гɶԵ��ӣ�
��2��a���������д���O-O����C-C����Ϊ�Ǽ��Լ���C-O��C-H��Ϊ���Լ���
b�����б���̼ԭ�ӣ����������Ľṹ��
c��C=O�����Цм���
��3����NaHΪ���Ӿ��壬NaH������ÿ����������Χ��6���⸺���ӣ��������ⳤΪa����Naԭ�Ӽ���С�˼��Ϊ$\frac{\sqrt{2}}{2}$a��
��B��OCH3��3��B��3��Oԭ�ӳɼ���B��OCH3��3����ͬ�ռ乹��ƽ�������Σ�
��NaBH4�������Ӽ�����λ�����ۼ���
��� �⣺��1����������ص�����Ԫ��ΪH��C��O��Ԫ�صķǽ�����Խǿ���縺��Խǿ���ǽ����ԣ�O��C��H����H��C��O����Ԫ�صĵ縺���ɴ�С��˳����O��C��H�����ڻ�̬Oԭ���У�������8�ĵ��ӣ�������3������гɶԵ��ӣ���������3�������෴�ĵ��ӣ�
�ʴ�Ϊ��O��C��H��3��
��2��a���������д���O-O����C-C����Ϊ�Ǽ��Լ���C-O��C-H��Ϊ���Լ�����a��ȷ��
b�����б���̼ԭ�ӣ����������Ľṹ��������̼ԭ�Ӳ�����ͬһƽ�棬��b����
c��C=O�����Цм�����c����
�ʴ�Ϊ��a��
��3����NaHΪ���Ӿ��壬NaH������ÿ����������Χ��6���⸺���ӣ��������ⳤΪa����Naԭ�Ӽ���С�˼��Ϊ$\frac{\sqrt{2}}{2}$a��
�ʴ�Ϊ�����ӣ� 6��$\frac{\sqrt{2}}{2}$a��
��B��OCH3��3��B��3��Oԭ�ӳɼ���Ϊsp2�ӻ���B��OCH3��3����ͬ�ռ乹��ƽ�������Σ���B��OCH3��3������ͬ�ռ乹�͵ķ��ӻ����ӿ�ΪSO3��CO32-��
�ʴ�Ϊ��sp2��SO3��CO32-��
��Bԭ�Ӻ��������3�����ӣ�NaBH4�������Ӽ�����λ�����ۼ����ʴ�Ϊ�����Ӽ�����λ�������ۼ���
���� ���⿼���Ϊ�ۺϣ��漰�л���Ľṹ�����ʣ��Լ������ļ��㡢���ӹ��͵�֪ʶ��Ϊ�߿��������ͺ�Ƶ���㣬������ѧ���ķ��������������Ŀ��飬�ѶȲ���

 ��У����ϵ�д�
��У����ϵ�д�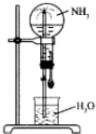 �ĸ��������Բ����ƿ�ֱ������NH3��HCl��NO2��NO2��O2�Ļ���������Ȫʵ�飬��ͼ��ʾ������ַ�Ӧ��ƿ����Һ�����ʵ���Ũ�ȴ�С��ϵΪ��������
�ĸ��������Բ����ƿ�ֱ������NH3��HCl��NO2��NO2��O2�Ļ���������Ȫʵ�飬��ͼ��ʾ������ַ�Ӧ��ƿ����Һ�����ʵ���Ũ�ȴ�С��ϵΪ��������| A�� | �٣��ڣ��ۣ��� | B�� | ��=��=��=�� | C�� | ��=��=�ۣ��� | D�� | ��=�ڣ��ۣ��� |
��֪������������������������ʽ��ʼ��������ȫ����ʱ��Һ��pH���±�
| ������ | Cu��OH��2 | Fe��OH��3 | Fe��OH��2 |
| ��ʼ����pH | 4.7 | 2.7 | 7.6 |
| ��ȫ����pH | 6.7 | 3.7 | 9.6 |
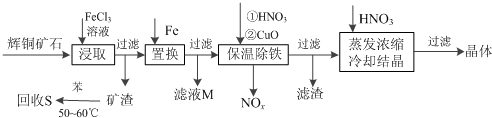
��2������NOx ��������Ϻ�ͨ��ˮ�������������п�ѭ�����õ�һ�����ʣ��÷�Ӧ�Ļ�ѧ����ʽΪ4NOx+��5-2x��O2+2H2O=4HNO3
��3��������ҺM�м��루��ͨ�룩����D������ĸ�����ʣ��õ��϶����һ�ֿ�ѭ�����õ����ʣ�
A��Fe B��NaClO C��H2O2 D��Cl2
��4������Ũ��ʱ��Ҫ�����������Һ��pH���������ǵ�����ҺpHʹʹ��Ԫ����ȫת��Ϊ������������������ͭ����ˮ�⣮
��5���ڳ����Σ����pH���Ʋ�����ʹ�������ͣ�����ΪӦ�ò�ȡ�IJ��ȴ�ʩ�ǣ����������뵽HNO3�н�pH��Ϊ3.7-4.7����ַ�Ӧ����ˣ�����Һ��ԭ��Һ�ϲ���
| A�� | ��NaOH��Һ | B�� | ���� | C�� | ��ϡ���� | D�� | ��Ca��OH��2��Һ |
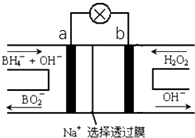 ���⻯��NaBH4��BԪ�صĻ��ϼ�Ϊ+3�ۣ�ȼ�ϵ�أ�DBFC�������ھ��б������ߡ������������Ⱦ��ȼ�����ڴ����������ŵ㣬����Ϊ��һ�ֺ��з�չDZ����ȼ�ϵ�أ��乤��ԭ����ͼ��ʾ������˵����ȷ���ǣ�������
���⻯��NaBH4��BԪ�صĻ��ϼ�Ϊ+3�ۣ�ȼ�ϵ�أ�DBFC�������ھ��б������ߡ������������Ⱦ��ȼ�����ڴ����������ŵ㣬����Ϊ��һ�ֺ��з�չDZ����ȼ�ϵ�أ��乤��ԭ����ͼ��ʾ������˵����ȷ���ǣ�������| A�� | �ŵ�ʱ��ÿת��2mol���ӣ���������Ҫ����9.5gNaBH4 | |
| B�� | �缫a����MnO2��MnO2�����缫�������д����� | |
| C�� | ��طŵ�ʱNa+��b��������a���� | |
| D�� | ��صĸ�����ӦΪBH4-+2H2O-8e-�TBO2-+8H+ |
| A�� | �⻯��ķе㣺Y��W | |
| B�� | �����ӵİ뾶��W��Y��Z��X | |
| C�� | W���������Ӧ��ˮ������һ��ǿ�� | |
| D�� | ��Zͬ�����һ����Ԫ�صĵ��ʲ�����N2��Ӧ |
| A�� | �ڹ���������ˮ�ķ�Ӧ�У�ÿ����0.1mol������ת�Ƶĵ�����ĿΪ0.4NA | |
| B�� | ����ȼ�ϵ����������22.4L����ʱ����·��ͨ���ĵ�����ĿΪ4NA | |
| C�� | ��30g�������辧���к��еĹ��ۼ���ĿΪ2NA | |
| D�� | �������º�NA��NO2��N2O4���ӵĻ��������ȴ����״���������ԼΪ22.4L |
| A�� | �����£����ȶ����ڵ�R�������ﶼ������������ | |
| B�� | R����̬�⻯���ˮ��Һ����ǿ���� | |
| C�� | R�ǽ���Ԫ�� | |
| D�� | R����̬�����ﲻ����������ȼ�� |
| A�� | ��ɫ��ѧ�ĺ����Ǵ�Դͷ�Ϸ�ֹ���������������Ի�������Ⱦ | |
| B�� | ���ٻ�ʯȼ�ϵ�ʹ�ã������ڽ��Ϳ�����pM2.5����ֵ | |
| C�� | ���ع��͡���ֹʳ�ã������������Ʋ��͡������� | |
| D�� | ���ۡ����͡������ʶ�����Ȼ�߷��ӻ����� |