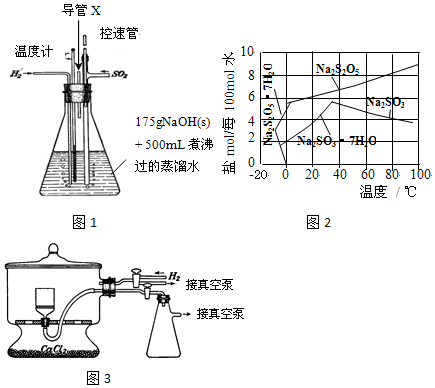
��Ŀ����
9��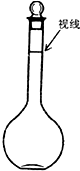 ʵ��������500mL��0.2mol/L��Na2SO4��Һ��ʵ����������У�
ʵ��������500mL��0.2mol/L��Na2SO4��Һ��ʵ����������У�������ƽ�ϳƳ�14.2g�����ƹ��壬���������ձ��У�������������ˮʹ����ȫ�ܽ⣮
�ڰ��Ƶõ���ҺС�ĵ�ע��500ml����ƿ�У�
�ۼ���������ƿ�м�����ˮ��Һ���̶�1cm��2cm�������ý�ͷ�ι�С�ĵμ�����ˮ����Һ����ײ���̶������У�
������������ˮϴ�ձ��Ͳ�����2-3�Σ�ÿ��ϴ�ӵ�Һ�嶼С��ע������ƿ��������ҡ�ȣ�
�ݽ�����ƿ�����������ҡ�ȣ�
����д���пհף�
��1�������������ȷ˳��Ϊ������ţ��٢ڢܢۢݣ�
��2����ʵ���õ��Ļ������������ձ�����ƽ�����롢���ӣ�����Ͳ����ȱ�ٵ������Dz�������500mL����ƿ����ͷ�ιܣ�
���� ��1���ù�������һ�����ʵ���Ũ����Һ��һ�㲽��Ϊ�����㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ����ǩ���ݴ�����
��2���������ƶ������ʵ���Ũ����Һһ�㲽��ѡ����Ҫ��������
��� �⣺��1���ù�������һ�����ʵ���Ũ����Һ��һ�㲽��Ϊ�����㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ����ǩ��������ȷ��˳��Ϊ���٢ڢܢۢݣ�
�ʴ�Ϊ���٢ڢܢۢݣ�
��2���ù�������һ�����ʵ���Ũ����Һ��һ�㲽��Ϊ�����㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ����ǩ���õ���������������ƽ��ҩ�ס��ձ�����������500mL����ƿ����ͷ�ιܣ�
���Ի�ȱ�ٵ���������������500mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ����������500mL����ƿ����ͷ�ιܣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ����ԭ�������������ǽ���ؼ���ע������ƿ����ѡ����Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ����������ϵ�д�
����������ϵ�д� �Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�����Ŀ
19��һ���¶�ʱ�����ݻ�Ϊ2L���ܱ������г���һ������SO2��O2��������Ӧ��2SO2��g��+O2��g��?2SO3��g������Ӧ�����вⶨ�IJ������ݼ��±�������t1��t2��������˵����ȷ���ǣ�������
| ��Ӧʱ��/min | n��SO2��/mol | n��O2��/mol |
| 0 | 0.10 | 0.060 |
| t1 | 0.012 | |
| t2 | 0.016 |
| A�� | ��Ӧ��0��t1 min�ڵ�ƽ������Ϊv��SO3��=0.088/t1 mol•L-1•min-1 | |
| B�� | ���������������䣬��ʼʱ�������г���0.10molSO3��0.010 molO2������ƽ��ʱ��n��SO2��=0.012 mol | |
| C�� | ���������������䣬�����¶ȣ�ƽ��ʱc��SO2��=0.0070mol•L-1����Ӧ�ġ�H��0 | |
| D�� | ��ͬ�¶��£���ʼʱ�������г���0.050molSO2��0.030molO2���ﵽƽ��ʱSO2ת���ʴ���88% |
17�����л�ѧ�����У������߱����ǣ�������
| A�� | �����ڿ�����ȼ�� | B�� | ��Br2����ȡ����Ӧ | ||
| C�� | ��H2�����ӳɷ�Ӧ | D�� | ��ʹ���������Һ��ɫ |
 �����������[HPO��OC2H5��2]Ϊ��Ҫ����ȼ���ܼ���ʵ���Ҳ������Ȼ�����ˮ�Ҵ��Ʊ��������������PCl3+4C2H5OH��HPO��OC2H5��2+HCl��+2C2H5Cl+H2O��ʵ�鲽�����£�
�����������[HPO��OC2H5��2]Ϊ��Ҫ����ȼ���ܼ���ʵ���Ҳ������Ȼ�����ˮ�Ҵ��Ʊ��������������PCl3+4C2H5OH��HPO��OC2H5��2+HCl��+2C2H5Cl+H2O��ʵ�鲽�����£�