��Ŀ����
4�����й��ڵ������Һ����������ȷ���ǣ�������| A�� | c��H+������1��10-7 mol•L-1����Һһ����������Һ | |
| B�� | 0.2 mol•L-1 CH3COOH��Һ�е�c��H+����0.1 mol•L-1 CH3COOH��Һ�е�c��H+����2�� | |
| C�� | ����ʱ��pH=2��CH3COOH��0.01 mol•L-1 NaOH��Һ�������ϣ���Ϻ�pH=7 | |
| D�� | �����ʵ���Ũ�ȵ�Na2S��NaHS�����Һ�У�2c��Na+��=3[c��S2-��+c��HS-��+c��H2S��] |
���� A����Һ�������ȡ���������Ӻ�������Ũ�ȵ���Դ�С��
B�����ݴ���Ũ��������̶ȼ�С���з�����
C��pH=2��CH3COOH��Һ�У�������Ũ��Ϊ0.01mol/L��
D�����ݵ�Ũ�ȡ�����������ơ����⻯�ƵĻ��Һ�е������غ�����жϣ�
��� �⣺A����Һ�������ȡ����������Ũ�Ⱥ�����������Ũ�ȵ���Դ�С��c��H+����c��OH-����Һ�����ԣ�c��H+��=c��OH-����Һ�����ԣ�c��H+����c��OH-����Һ�ʼ��ԣ���A����
B��������������ʣ�Ũ��������ĵ���̶ȼ�С������0.2 mol/L CH3COOH��Һ�е�H+Ũ��С��0.1 mol/L CH3COOH��Һ�е�H+Ũ�ȵ�2������B����
C��pH=2��CH3COOH��Һ�У�������Ũ��Ϊ0.01mol/L�������Ũ�ȴ���0.01mol/L���ʺ�0.01mol/L������������Һ�������Ϻ����������Һ�����ԣ���pH��7����C����
D��0.1mol•L-1Na2S��Һ��0.1mol•L-1 NaHS��Һ�������ϣ����������غ�ɵã�2c��Na+��=3c��S2-��+3c��HS-��+3c��H2S���������ɵã�2c��Na+��-3c��S2-��=3c��HS-��+3c��H2S������D��ȷ��
��ѡD��
���� ���⿼������Һ����Ե��жϡ���Һ��������Ũ�ȵĴ�С�ȽϺ�����Һ�е������غ㣬�ۺ��Խ�ǿ��ע����������գ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ���淴Ӧ������ʱ��ij����� | |
| B�� | ���淴Ӧ�����Dz��ɸı�� | |
| C�� | �κο��淴Ӧ�ڸ����������¶���һ������ | |
| D�� | ��ͬ�Ŀ��淴Ӧ������ͬ��������һ�㲻ͬ |
| A�� | K+��Cu2+��Cl-��OH- | B�� | Mg2+��SO42-��K+��Cl- | ||
| C�� | Na+��H+��NO3-��CO32- | D�� | Ba2+��Na+��OH-��SO42- |
| A�� | 1��1��1 | B�� | 1��4��2 | C�� | 1��2��4 | D�� | 1��3��2 |
| A�� | N2 | B�� | Cl2 | C�� | NH3 | D�� | SO2 |
| A�� | Mgʧ������CO2���Naʧ��Ҳ������CO2��� | |
| B�� | Fe3O4��д��FeO•Fe2O3��Pb3O4Ҳ��д��PbO•Pb2O3 | |
| C�� | Cu��Cl2ֱ�ӻ�������CuCl2��Cu��Sֱ�ӻ��ϵõ�CuS | |
| D�� | CO2ͨ�뵽���ᱵ��Һ�в������ɳ�����SO2ͨ�뵽���ᱵ��Һ��Ҳ�������ɳ��� |
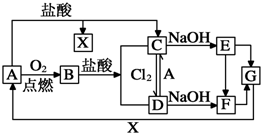 ��֪A��һ�ֳ���������F��һ�ֺ��ɫ�������Ը���ͼ��ת����ϵ���ش��������⣮
��֪A��һ�ֳ���������F��һ�ֺ��ɫ�������Ը���ͼ��ת����ϵ���ش��������⣮ ��
��