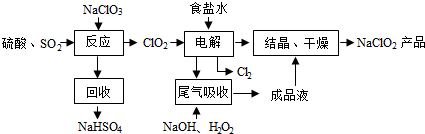
��Ŀ����
6��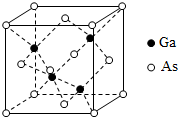 �黯�أ�GaAs���������İ뵼����ϣ������������ͼ�������̫���ܵ�صIJ��ϵȣ��ش��������⣺
�黯�أ�GaAs���������İ뵼����ϣ������������ͼ�������̫���ܵ�صIJ��ϵȣ��ش��������⣺��1��д����̬Asԭ�ӵĺ�������Ų�ʽ1s22s22p63s23p63d104s24p3��
��2������Ԫ�������ɣ�ԭ�Ӱ뾶Ga����As����һ������GaС��As��������ڡ���С�ڡ���
��3��AsCl3���ӵ����幹��Ϊ�����Σ�����As���ӻ��������Ϊsp3��
��4��GaF3���۵����1000�棬GaCl3���۵�Ϊ77.9�棬��ԭ����GaF3Ϊ���Ӿ��壬GaCl3Ϊ���Ӿ��壬���Ӿ�����۵�ߣ�
��5��GaAs���۵�Ϊ1238�棬�ܶ�Ϊ�� g•cm-3���侧���ṹ��ͼ��ʾ���þ��������Ϊԭ�Ӿ��壬Ga��As�Թ��ۼ����ϣ�Ga��As��Ħ�������ֱ�ΪMGa g•mol-1��MAs g•mol-1��ԭ�Ӱ뾶�ֱ�ΪrGa pm��rAs pm������٤������ֵΪNA����GaAs������ԭ�ӵ����ռ��������İٷ���Ϊ$\frac{4�С�1{0}^{-30}N{\;}_{A}�ѣ�{r}_{Ga}^{3}+{r}_{As}^{3}��}{3��{M}_{Ga}+{M}_{As}��}$��100%��
���� ��1��AsΪ��A��33��Ԫ�أ������Ų�ʽΪ��1s22s22p63s23p63d104s24p3��
��2��ͬһ���ڣ�ԭ������ԽС�뾶Խ��ͬ���ڵ�һ�����ܴ����ң�������
��3��AsCl3�м۲���ӶԸ���=�Ҽ�����+�µ��ӶԸ���=3+$\frac{5-3��1}{2}$=4������ԭ���ӻ���ʽ��sp3��������һ�Թ¶Ե��Ӷԣ����ӿռ乹��Ϊ�����Σ�
��4��GaF3���۵����1000�棬GaCl3���۵�Ϊ77.9�棬��ԭ����GaF3Ϊ���Ӿ��壬GaCl3Ϊ���Ӿ��壬���Ӿ�����۵�ߣ�
��5��GaAs���۵�Ϊ1238�棬�۵�ϸߣ��Թ��ۼ�����γ�����ԭ�Ӿ��壬�ܶ�Ϊ�� g•cm-3�����ݾ�̯�����㣬As��$8��\frac{1}{8}+6��\frac{1}{2}=4$��Ga��4��1=4�����侧����ԭ����ռ�����V1=��$\frac{4}{3}��{{r}^{3}}_{As}��4+\frac{4}{3}��{{r}^{3}}_{Ga}��4$����10-30�����������V2=$\frac{m}{��}$=$\frac{4��\frac{��{M}_{Ga}+{M}_{As}��}{{N}_{A}}}{��}$����GaAs������ԭ�ӵ����ռ��������İٷ���Ϊ$\frac{{V}_{1}}{{V}_{2}}��100%$��V1��V2�������ðٷ���=$\frac{4�С�1{0}^{-30}N{\;}_{A}�ѣ�{r}_{Ga}^{3}+{r}_{As}^{3}��}{3��{M}_{Ga}+{M}_{As}��}$��100%��
��� �⣺��1��AsΪ��A��33��Ԫ�أ������Ų�ʽΪ��1s22s22p63s23p63d104s24p3���ʴ�Ϊ��1s22s22p63s23p63d104s24p3��
��2������Ԫ�������ɣ�Ga��Asλ��ͬһ���ڣ�Gaԭ������С��As���ʰ뾶Ga����As��ͬ���ڵ�һ�����ܴ����ң������ʵ�һ������GaС��As��
�ʴ�Ϊ�����ڣ�С�ڣ�
��3��AsCl3�м۲���ӶԸ���=�Ҽ�����+�µ��ӶԸ���=3+$\frac{5-3��1}{2}$=4������ԭ���ӻ���ʽ��sp3��������һ�Թ¶Ե��Ӷԣ����ӿռ乹��Ϊ�����Σ�
�ʴ�Ϊ�������Σ�sp3��
��4��GaF3���۵����1000�棬GaCl3���۵�Ϊ77.9�棬��ԭ����GaF3Ϊ���Ӿ��壬GaCl3Ϊ���Ӿ��壬���Ӿ�����۵�ߣ�
�ʴ�Ϊ��GaF3Ϊ���Ӿ��壬GaCl3Ϊ���Ӿ��壬���Ӿ�����۵�ߣ�
��5��GaAs���۵�Ϊ1238�棬�۵�ϸߣ��Թ��ۼ�����γ�����ԭ�Ӿ��壬�ܶ�Ϊ�� g•cm-3�����ݾ�̯�����㣬As��$8��\frac{1}{8}+6��\frac{1}{2}=4$��Ga��4��1=4�����侧����ԭ����ռ�����V1=��$\frac{4}{3}��{{r}^{3}}_{As}��4+\frac{4}{3}��{{r}^{3}}_{Ga}��4$����10-30�����������V2=$\frac{m}{��}$=$\frac{4��\frac{��{M}_{Ga}+{M}_{As}��}{{N}_{A}}}{��}$����GaAs������ԭ�ӵ����ռ��������İٷ���Ϊ$\frac{{V}_{1}}{{V}_{2}}��100%$��V1��V2�������ðٷ���=$\frac{4�С�1{0}^{-30}N{\;}_{A}�ѣ�{r}_{Ga}^{3}+{r}_{As}^{3}��}{3��{M}_{Ga}+{M}_{As}��}$��100%��
�ʴ�Ϊ��ԭ�Ӿ��壻���ۣ�$\frac{4�С�1{0}^{-30}N{\;}_{A}�ѣ�{r}_{Ga}^{3}+{r}_{As}^{3}��}{3��{M}_{Ga}+{M}_{As}��}$��100%��
���� ���⿼���˷��ӿռ乹�͡������Ų�ʽ��ԭ���ӻ���ʽ�������ܶȵļ��㡢�����ܼ��뾶��С�Ƚϵ�֪ʶ���ۺ��Խ�ǿ�����ļ����ѶȽϴ�Ҫ��ѧ���н��Ͻ���̬�Ⱥ���ʵ�Ļ�����Ҳ�Ƕ�ѧ�������Ŀ��飮

| A�� | ����Һ��ϡ�����У�����Ũ���еĿ��������еĿ��ܼ�С���еĿ��ܲ��� | |
| B�� | ��AlCl3��Һ��Na2SO3��Һ���ɲ����տɷֱ�õ�Al2O3��Na2SO3 | |
| C�� | 25��ʱ����pH��ͬ��ϡ���������ֱ��к�ͬŨ�ȡ�ͬ�����NaOH��Һ�������Һ������ʱ������ϡ���������������ͬ | |
| D�� | 25��ʱ��pH=9��NH4Cl��NH3•H2O�����Һ��c��Cl-����c��NH4+�� |
| ѡ�� | n��CO2��/mol | ��Һ�����ӵ����ʵ���Ũ�� |
| A | 0 | c��Na+����c��AlO2-��+c��OH-�� |
| B | 0.01 | c��Na+����c��AlO2-����c��OH-����c��CO32-�� |
| C | 0.015 | c��Na+����c��HCO3-����c��CO32-����C��OH-�� |
| D | 0.03 | c��Na+����c��HCO3-����c��OH-����c��H+�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| ʵ�� | ���� | |
| A | ����ϡ���� | �Ƚ�Ũ��������ձ��У���������ˮ |
| B | ��ˮ���ռ�KMnO4�ֽ������O2 | ��Ϩ��ƾ��ƣ����Ƴ����� |
| C | Ũ������MnO2��Ӧ�Ʊ�����Cl2 | ���������ͨ��Ũ���ᣬ��ͨ������ʳ��ˮ |
| D | CCl4��ȡ��ˮ�е�I2 | �ȴӷ�Һ©���¿ڷų��л��㣬����Ͽڵ���ˮ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | pH=10��NaOH��Һ��pH=4��������Һ�У�ˮ�ĵ���̶� | |
| B�� | ��pH=3������ʹ���ֱ�ϡ�ͳ�pH=5����Һ�����ˮ����� | |
| C�� | 10mL0.1mol•L-1�Ĵ�����100mL0.01mol•L-1�Ĵ����е�H+���ʵ��� | |
| D�� | �����pH��ȵĴ�������ᣬ�ֱ��������п�۷�Ӧ������H2������ |
| A�� | ��ͭ����ϡ�����У�Cu+4H++2NO3-�TCu2++2NO2��+H2O | |
| B�� | ��Fe2��SO4��3��Һ�м���������ۣ�Fe3++Fe�T2Fe2+ | |
| C�� | ��Al2��SO4��3��Һ�м��������ˮ��Al3++3NH3•H2O�TAl��OH��3��+3NH4+ | |
| D�� | ��Na2SiO3��Һ�еμ�ϡ���Na2SiO3+3H+�TH2SiO3��+3Na+ |