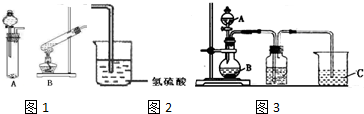
��Ŀ����
15���١�=10 ���Ǽ����л�������ơ�����ʽ��ṹ��ʽ����C2H2 �������� �۱� ��
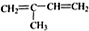 ��
��
��CH3CH��C2H5��CH2CH��C2H5��CH3 ��C5H10 ��ClCH=CHCl ��C5H4
��
�ݴ˻ش��������⣺
��1��ʵ�����ƢٵĻ�ѧ����ʽ��CaC2+2H2O��Ca��OH��2+C2H2����
�����ƣ�3��5-�������飮
��2���۵�ͬϵ��A�������й���66�����ӣ�A������һ�����ֻ��һ�֣�A�Ľṹ��ʽ
 ��
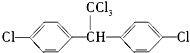
����3��ij��B����14.3%���ҷ�������������ȫ��Ч����80g��Br25%����ˮ�м�����л����ˮ�պ���ȫ��ɫ����ʱҺ��������82.1g����B�Ľṹ��ʽΪ����CH3��2C=C��CH3��2��
��4����������ͬ���칹�����Ŀ������30���֣��磺A��CH2=C=C=C=CH2 B��CH��C-CH=C=CH2 C��
 D��
D��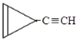 ������A��B����״���ӣ���������Щ�ṹ�ܷ��ȶ����ڣ�����д������̼ԭ�Ӿ����ߵ�һ����״���ӵĽṹ��ʽ��CH��C-C��C-CH3��
������A��B����״���ӣ���������Щ�ṹ�ܷ��ȶ����ڣ�����д������̼ԭ�Ӿ����ߵ�һ����״���ӵĽṹ��ʽ��CH��C-C��C-CH3����5��������������23��ԭ�ӹ��森
���� ��1��ʵ������̼���ƺ�ˮ��Ӧ�Ʊ���Ȳ��������Ϊ7��̼ԭ�ӣ�����2������
��2������A�DZ���ͬϵ����ڽṹ��ֻ��һ���������Ҳ���Ϊ��������������ͨʽCnH2n-6�����ڵ�����Ϊ66�����У�6n+2n-6=66�����n=9����A�ķ���ʽΪC9H20������A�ı�����һ�����ֻ��һ�֣���A�Ľṹ�ܶԳƣ�
��3��������������ֻ��C��H����Ԫ�أ���ij��B����14.3%����̼85.7%���ʴ��л����е�C��Hԭ�Ӹ���֮��Ϊ��$\frac{14.3%}{1}��\frac{85.7%}{12}$=1��2�����л���ķ���ʽΪ��CH2��n����ˮ��������2.1g��Ϊ��ˮ���յĸ��������������ʵ���n=$\frac{2.1g}{14ng/mol}$=$\frac{3}{20n}$mol����80g��Br5%����ˮ��������ʵ���Ϊn=$\frac{80g��5%}{160g/mol}$=0.025mol������1mol��CH2��n��1mol�壬���У�$\frac{3}{20n}$=0.025�����n=6�����ڷ�������������ȫ��Ч����B�Ľṹ��ʽΪ����CH3��2C=C��CH3��2��
��4����C5H4�IJ����Ͷȣ�Ϊ4����̼ԭ�Ӿ�����ʱ��Ӧ��������ȲΪĸ��Ľṹ������һ��̼̼�����IJ����Ͷ�Ϊ2����������������л����к�2��̼̼������
��5������Ϊƽ���νṹ���뱽��ֱ��������ԭ����ͬһ��ƽ���ϣ���������ȷ��һ��ƽ���֪���������������е�ԭ���Լ����ӱ�����Cԭ�ӿ��ܹ�ƽ�棬��23��ԭ�ӣ�
��� �⣺��1��ʵ������̼���ƺ�ˮ��Ӧ�Ʊ���Ȳ������ʽΪCaC2+2H2O��Ca��OH��2+C2H2����������Ϊ3��5-�������飬
�ʴ�Ϊ��CaC2+2H2O��Ca��OH��2+C2H2����3��5-�������飻
��2������A�DZ���ͬϵ����ڽṹ��ֻ��һ���������Ҳ���Ϊ��������������ͨʽCnH2n-6�����ڵ�����Ϊ66�����У�6n+2n-6=66�����n=9����A�ķ���ʽΪC9H20������A�ı�����һ�����ֻ��һ�֣���A�Ľṹ�ܶԳƣ��ṹΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3��������������ֻ��C��H����Ԫ�أ���ij��B����14.3%����̼85.7%���ʴ��л����е�C��Hԭ�Ӹ���֮��Ϊ��$\frac{14.3%}{1}��\frac{85.7%}{12}$=1��2�����л���ķ���ʽΪ��CH2��n����ˮ��������2.1g��Ϊ��ˮ���յĸ��������������ʵ���n=$\frac{2.1g}{14ng/mol}$=$\frac{3}{20n}$mol����80g��Br5%����ˮ��������ʵ���Ϊn=$\frac{80g��5%}{160g/mol}$=0.025mol������1mol��CH2��n��1mol�壬���У�$\frac{3}{20n}$=0.025�����n=6�����ڷ�������������ȫ��Ч����B�Ľṹ��ʽΪ����CH3��2C=C��CH3��2���ʴ�Ϊ����CH3��2C=C��CH3��2��
��4����C5H4�IJ����Ͷȣ�Ϊ4����̼ԭ�Ӿ�����ʱ��Ӧ��������ȲΪĸ��Ľṹ������һ��̼̼�����IJ����Ͷ�Ϊ2����������������л����к�2��̼̼�������ʴ��л���ĽṹΪCH��C-C��C-CH3���ʴ�Ϊ��CH��C-C��C-CH3��
��5������Ϊƽ���νṹ���뱽��ֱ��������ԭ����ͬһ��ƽ���ϣ���������ȷ��һ��ƽ���֪���������������е�ԭ���Լ����ӱ�����Cԭ�ӿ��ܹ�ƽ�棬��23��ԭ�ӣ��ʴ�Ϊ��23��
���� �����ۺϿ����л���Ľṹ�����ʣ�Ϊ��Ƶ���㣬����ѧ���ķ��������Ŀ��飬ע������л���Ľṹ�Լ�ͬ���칹����жϣ�Ϊ������ѵ���״��㣬��Ŀ�Ѷ��еȣ�

| A�� | ��������ѹǿ | B�� | �������������ʵ��� | ||
| C�� | ��������ƽ����Է������� | D�� | v��B��=3V��D�� |
| A�� | BeCl2 | B�� | H2O2 | C�� | COCl2 | D�� | SF6 |
| A�� | 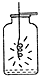 ��˿ȼ�� | B�� | 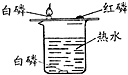 ����ȼ�� | C�� | 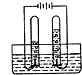 ���ˮ | D�� | 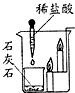 �����Ϩ�� |
 _��
_��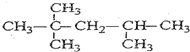 ��
��